(primary solid tumor cohort)
This pipeline computes the correlation between cancer subtypes identified by different molecular patterns and selected clinical features.
Testing the association between subtypes identified by 10 different clustering approaches and 4 clinical features across 451 patients, 21 significant findings detected with P value < 0.05 and Q value < 0.25.
-
CNMF clustering analysis on array-based mRNA expression data identified 4 subtypes that correlate to 'AGE' and 'HISTOLOGICAL.TYPE'.
-
Consensus hierarchical clustering analysis on array-based mRNA expression data identified 3 subtypes that correlate to 'HISTOLOGICAL.TYPE'.
-
5 subtypes identified in current cancer cohort by 'CN CNMF'. These subtypes correlate to 'Time to Death', 'AGE', and 'HISTOLOGICAL.TYPE'.
-
3 subtypes identified in current cancer cohort by 'METHLYATION CNMF'. These subtypes correlate to 'HISTOLOGICAL.TYPE'.
-
CNMF clustering analysis on RPPA data identified 6 subtypes that correlate to 'HISTOLOGICAL.TYPE'.
-
Consensus hierarchical clustering analysis on RPPA data identified 6 subtypes that correlate to 'HISTOLOGICAL.TYPE'.
-
CNMF clustering analysis on sequencing-based mRNA expression data identified 3 subtypes that correlate to 'Time to Death', 'AGE', and 'HISTOLOGICAL.TYPE'.
-
Consensus hierarchical clustering analysis on sequencing-based mRNA expression data identified 4 subtypes that correlate to 'AGE', 'HISTOLOGICAL.TYPE', and 'RADIATIONS.RADIATION.REGIMENINDICATION'.
-
CNMF clustering analysis on sequencing-based miR expression data identified 3 subtypes that correlate to 'Time to Death', 'AGE', and 'HISTOLOGICAL.TYPE'.
-
Consensus hierarchical clustering analysis on sequencing-based miR expression data identified 3 subtypes that correlate to 'Time to Death', 'AGE', and 'HISTOLOGICAL.TYPE'.
Table 1. Get Full Table Overview of the association between subtypes identified by 10 different clustering approaches and 4 clinical features. Shown in the table are P values (Q values). Thresholded by P value < 0.05 and Q value < 0.25, 21 significant findings detected.
|
Clinical Features |
Time to Death |
AGE |
HISTOLOGICAL TYPE |
RADIATIONS RADIATION REGIMENINDICATION |
| Statistical Tests | logrank test | ANOVA | Chi-square test | Fisher's exact test |
| mRNA CNMF subtypes |
0.942 (1.00) |
0.0107 (0.215) |
0.000456 (0.0132) |
0.0317 (0.57) |
| mRNA cHierClus subtypes |
0.942 (1.00) |
0.0598 (0.897) |
0.00134 (0.0349) |
0.0666 (0.932) |
| CN CNMF |
0.00599 (0.132) |
5.7e-10 (1.94e-08) |
2.07e-35 (8.29e-34) |
0.0983 (1.00) |
| METHLYATION CNMF |
0.024 (0.456) |
0.0445 (0.757) |
2.17e-18 (8.03e-17) |
0.206 (1.00) |
| RPPA CNMF subtypes |
0.778 (1.00) |
0.343 (1.00) |
3.91e-06 (0.000117) |
0.327 (1.00) |
| RPPA cHierClus subtypes |
0.954 (1.00) |
0.463 (1.00) |
0.00421 (0.0968) |
0.584 (1.00) |
| RNAseq CNMF subtypes |
0.00308 (0.0739) |
1.1e-06 (3.53e-05) |
2.35e-23 (8.92e-22) |
0.0553 (0.885) |
| RNAseq cHierClus subtypes |
0.0985 (1.00) |
8.03e-07 (2.65e-05) |
2.96e-17 (1.03e-15) |
0.00715 (0.15) |
| MIRseq CNMF subtypes |
0.00052 (0.0146) |
1.13e-06 (3.53e-05) |
5.84e-26 (2.28e-24) |
0.406 (1.00) |
| MIRseq cHierClus subtypes |
0.00112 (0.0302) |
0.00244 (0.0611) |
2.28e-18 (8.21e-17) |
0.207 (1.00) |
Table S1. Get Full Table Description of clustering approach #1: 'mRNA CNMF subtypes'
| Cluster Labels | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| Number of samples | 13 | 19 | 14 | 8 |
P value = 0.942 (logrank test), Q value = 1
Table S2. Clustering Approach #1: 'mRNA CNMF subtypes' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 54 | 7 | 6.0 - 133.2 (35.4) |
| subtype1 | 13 | 2 | 9.0 - 133.2 (39.0) |
| subtype2 | 19 | 3 | 6.0 - 113.2 (37.7) |
| subtype3 | 14 | 1 | 8.6 - 89.3 (30.9) |
| subtype4 | 8 | 1 | 6.4 - 65.5 (22.9) |
Figure S1. Get High-res Image Clustering Approach #1: 'mRNA CNMF subtypes' versus Clinical Feature #1: 'Time to Death'
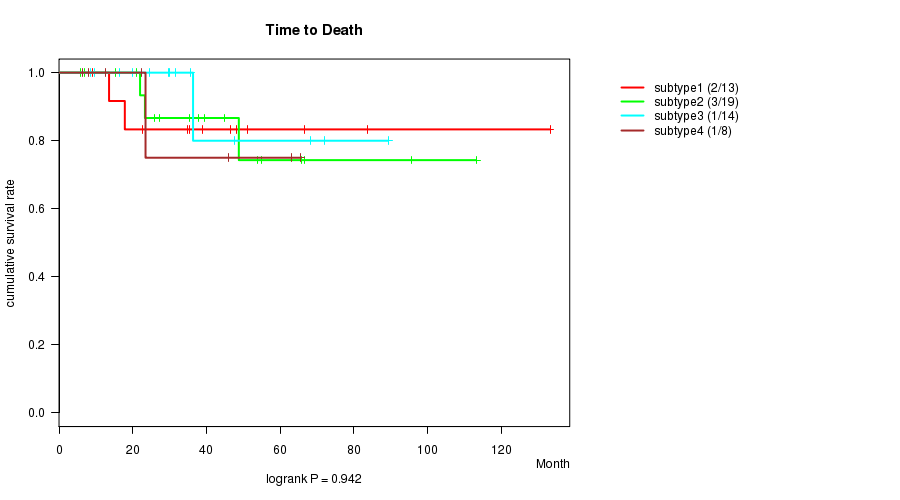
P value = 0.0107 (ANOVA), Q value = 0.21
Table S3. Clustering Approach #1: 'mRNA CNMF subtypes' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
| ALL | 54 | 62.9 (11.8) |
| subtype1 | 13 | 65.1 (12.0) |
| subtype2 | 19 | 68.4 (9.1) |
| subtype3 | 14 | 58.2 (11.0) |
| subtype4 | 8 | 54.8 (12.9) |
Figure S2. Get High-res Image Clustering Approach #1: 'mRNA CNMF subtypes' versus Clinical Feature #2: 'AGE'
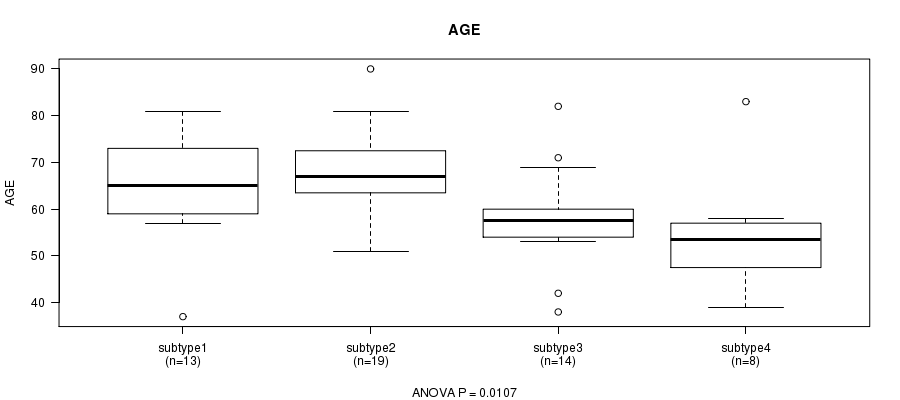
P value = 0.000456 (Chi-square test), Q value = 0.013
Table S4. Clustering Approach #1: 'mRNA CNMF subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|
| ALL | 41 | 1 | 12 |
| subtype1 | 12 | 0 | 1 |
| subtype2 | 8 | 0 | 11 |
| subtype3 | 13 | 1 | 0 |
| subtype4 | 8 | 0 | 0 |
Figure S3. Get High-res Image Clustering Approach #1: 'mRNA CNMF subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
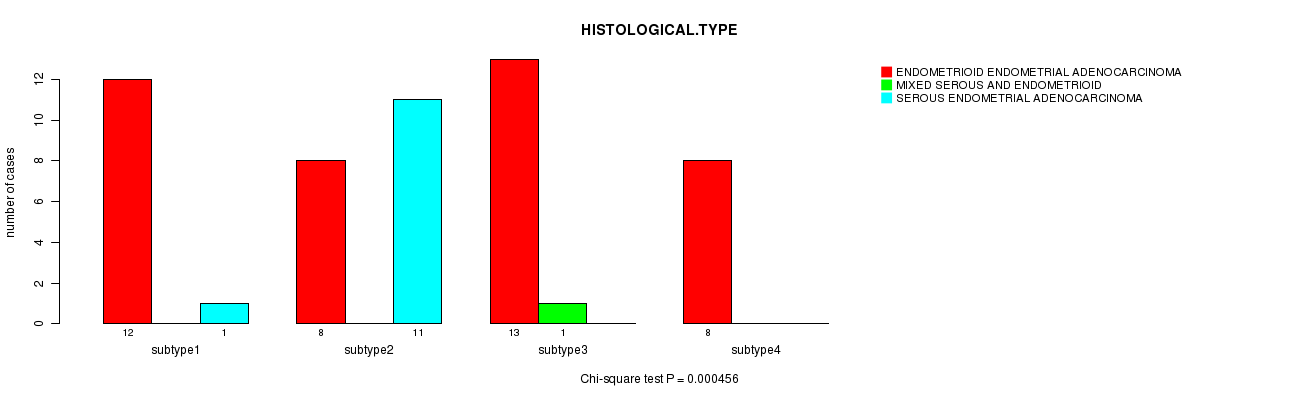
P value = 0.0317 (Fisher's exact test), Q value = 0.57
Table S5. Clustering Approach #1: 'mRNA CNMF subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 25 | 29 |
| subtype1 | 7 | 6 |
| subtype2 | 13 | 6 |
| subtype3 | 3 | 11 |
| subtype4 | 2 | 6 |
Figure S4. Get High-res Image Clustering Approach #1: 'mRNA CNMF subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
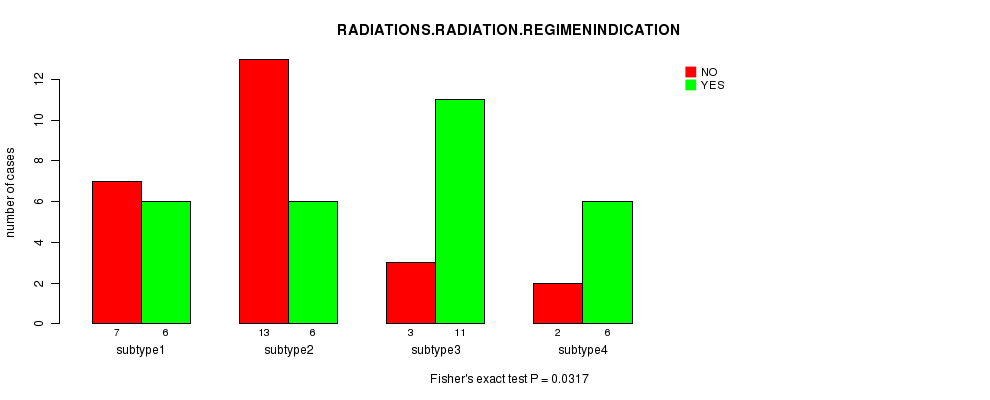
Table S6. Get Full Table Description of clustering approach #2: 'mRNA cHierClus subtypes'
| Cluster Labels | 1 | 2 | 3 |
|---|---|---|---|
| Number of samples | 20 | 15 | 19 |
P value = 0.942 (logrank test), Q value = 1
Table S7. Clustering Approach #2: 'mRNA cHierClus subtypes' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 54 | 7 | 6.0 - 133.2 (35.4) |
| subtype1 | 20 | 2 | 6.4 - 89.3 (29.8) |
| subtype2 | 15 | 2 | 9.0 - 133.2 (39.0) |
| subtype3 | 19 | 3 | 6.0 - 113.2 (37.7) |
Figure S5. Get High-res Image Clustering Approach #2: 'mRNA cHierClus subtypes' versus Clinical Feature #1: 'Time to Death'
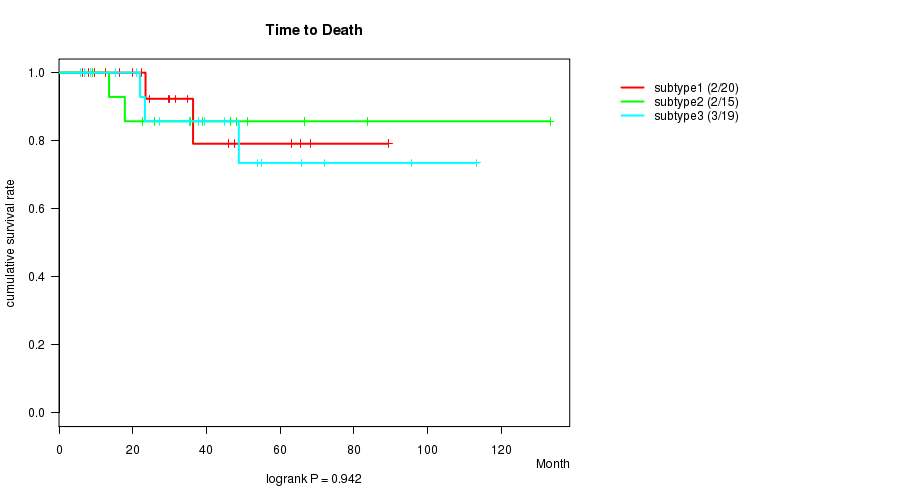
P value = 0.0598 (ANOVA), Q value = 0.9
Table S8. Clustering Approach #2: 'mRNA cHierClus subtypes' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
| ALL | 54 | 62.9 (11.8) |
| subtype1 | 20 | 58.2 (13.3) |
| subtype2 | 15 | 64.0 (10.6) |
| subtype3 | 19 | 67.1 (9.9) |
Figure S6. Get High-res Image Clustering Approach #2: 'mRNA cHierClus subtypes' versus Clinical Feature #2: 'AGE'
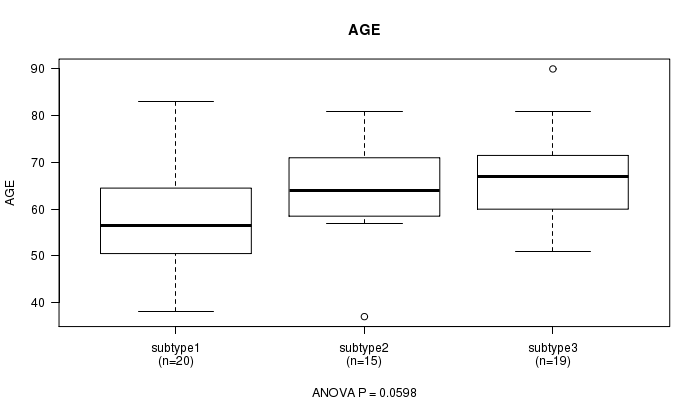
P value = 0.00134 (Chi-square test), Q value = 0.035
Table S9. Clustering Approach #2: 'mRNA cHierClus subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|
| ALL | 41 | 1 | 12 |
| subtype1 | 19 | 1 | 0 |
| subtype2 | 13 | 0 | 2 |
| subtype3 | 9 | 0 | 10 |
Figure S7. Get High-res Image Clustering Approach #2: 'mRNA cHierClus subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
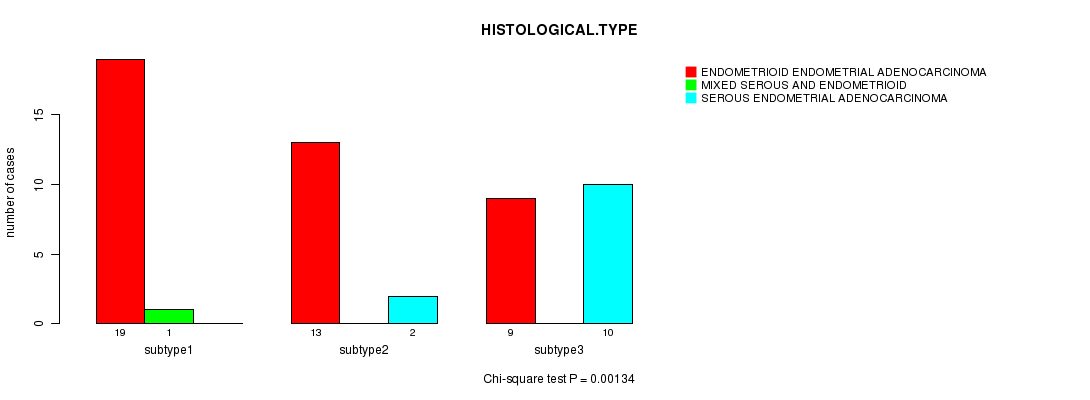
P value = 0.0666 (Fisher's exact test), Q value = 0.93
Table S10. Clustering Approach #2: 'mRNA cHierClus subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 25 | 29 |
| subtype1 | 5 | 15 |
| subtype2 | 9 | 6 |
| subtype3 | 11 | 8 |
Figure S8. Get High-res Image Clustering Approach #2: 'mRNA cHierClus subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
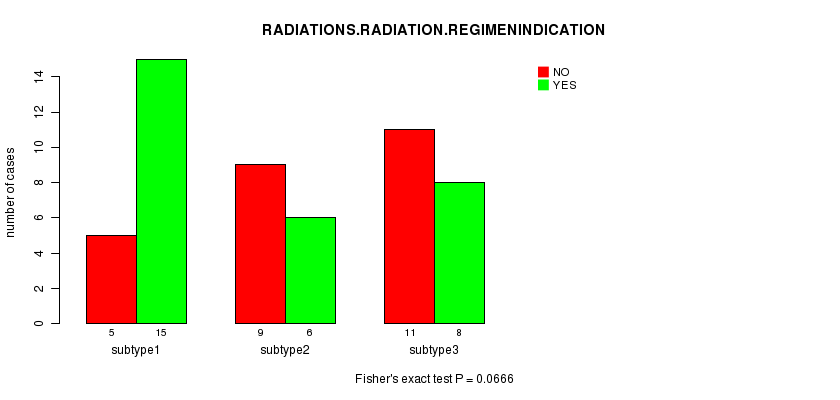
Table S11. Get Full Table Description of clustering approach #3: 'CN CNMF'
| Cluster Labels | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| Number of samples | 273 | 37 | 111 | 16 | 6 |
P value = 0.00599 (logrank test), Q value = 0.13
Table S12. Clustering Approach #3: 'CN CNMF' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 438 | 41 | 0.0 - 187.1 (15.8) |
| subtype1 | 270 | 18 | 0.0 - 187.1 (17.3) |
| subtype2 | 37 | 4 | 0.2 - 133.2 (8.0) |
| subtype3 | 109 | 15 | 0.0 - 113.2 (13.1) |
| subtype4 | 16 | 4 | 1.7 - 33.5 (13.9) |
| subtype5 | 6 | 0 | 0.3 - 31.3 (18.6) |
Figure S9. Get High-res Image Clustering Approach #3: 'CN CNMF' versus Clinical Feature #1: 'Time to Death'
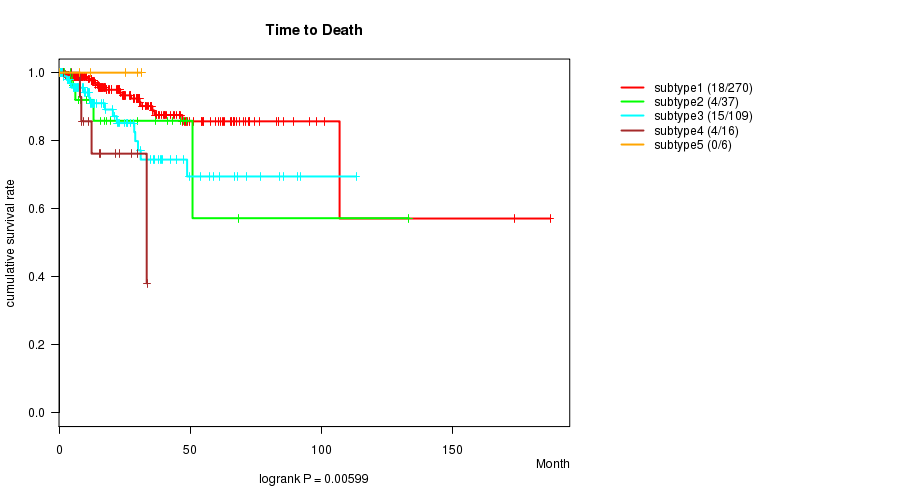
P value = 5.7e-10 (ANOVA), Q value = 1.9e-08
Table S13. Clustering Approach #3: 'CN CNMF' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
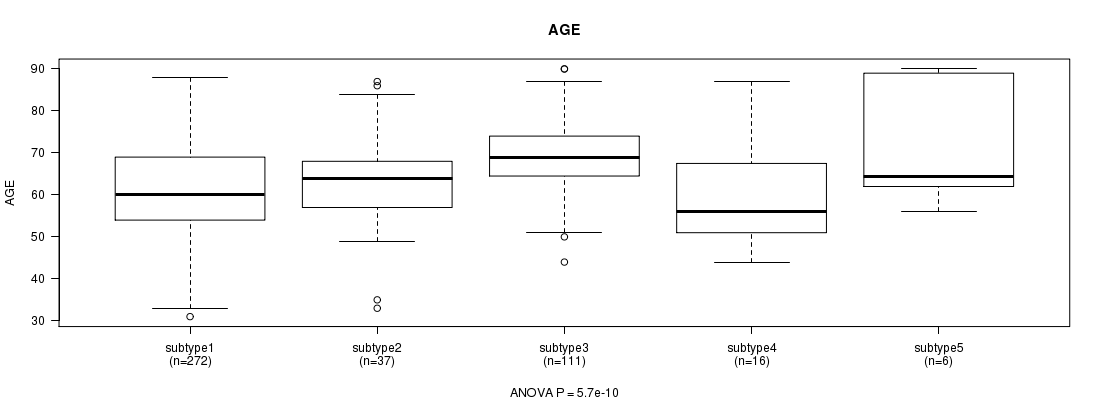
| ALL | 442 | 63.5 (11.2) |
| subtype1 | 272 | 61.2 (11.2) |
| subtype2 | 37 | 63.2 (11.9) |
| subtype3 | 111 | 69.5 (8.2) |
| subtype4 | 16 | 60.2 (12.9) |
| subtype5 | 6 | 71.0 (14.7) |
Figure S10. Get High-res Image Clustering Approach #3: 'CN CNMF' versus Clinical Feature #2: 'AGE'
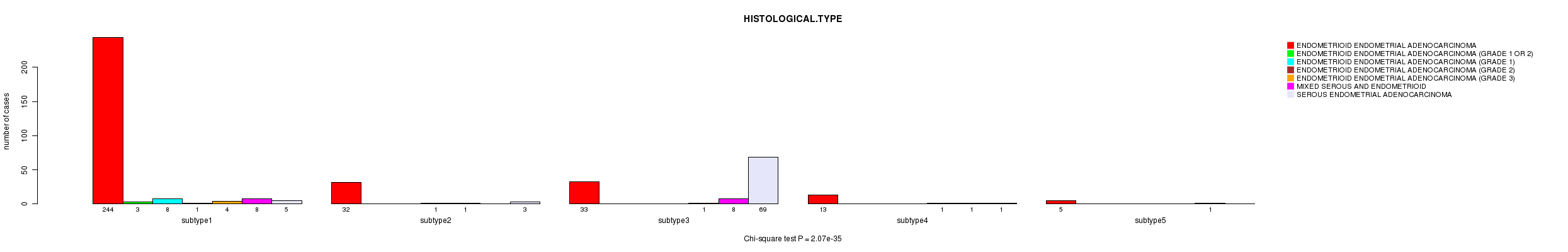
P value = 2.07e-35 (Chi-square test), Q value = 8.3e-34
Table S14. Clustering Approach #3: 'CN CNMF' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1 OR 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 3) | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|---|---|---|---|
| ALL | 327 | 3 | 8 | 2 | 7 | 18 | 78 |
| subtype1 | 244 | 3 | 8 | 1 | 4 | 8 | 5 |
| subtype2 | 32 | 0 | 0 | 1 | 1 | 0 | 3 |
| subtype3 | 33 | 0 | 0 | 0 | 1 | 8 | 69 |
| subtype4 | 13 | 0 | 0 | 0 | 1 | 1 | 1 |
| subtype5 | 5 | 0 | 0 | 0 | 0 | 1 | 0 |
Figure S11. Get High-res Image Clustering Approach #3: 'CN CNMF' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
P value = 0.0983 (Chi-square test), Q value = 1
Table S15. Clustering Approach #3: 'CN CNMF' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 133 | 310 |
| subtype1 | 94 | 179 |
| subtype2 | 6 | 31 |
| subtype3 | 27 | 84 |
| subtype4 | 4 | 12 |
| subtype5 | 2 | 4 |
Figure S12. Get High-res Image Clustering Approach #3: 'CN CNMF' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
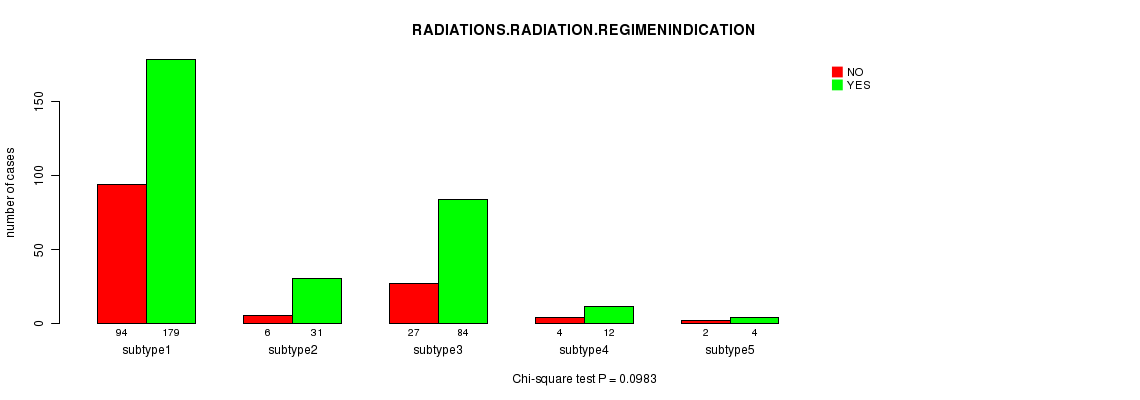
Table S16. Get Full Table Description of clustering approach #4: 'METHLYATION CNMF'
| Cluster Labels | 1 | 2 | 3 |
|---|---|---|---|
| Number of samples | 119 | 73 | 142 |
P value = 0.024 (logrank test), Q value = 0.46
Table S17. Clustering Approach #4: 'METHLYATION CNMF' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 329 | 31 | 0.0 - 187.1 (12.3) |
| subtype1 | 116 | 18 | 0.0 - 187.1 (11.4) |
| subtype2 | 73 | 4 | 0.0 - 92.0 (16.1) |
| subtype3 | 140 | 9 | 0.1 - 173.6 (11.8) |
Figure S13. Get High-res Image Clustering Approach #4: 'METHLYATION CNMF' versus Clinical Feature #1: 'Time to Death'
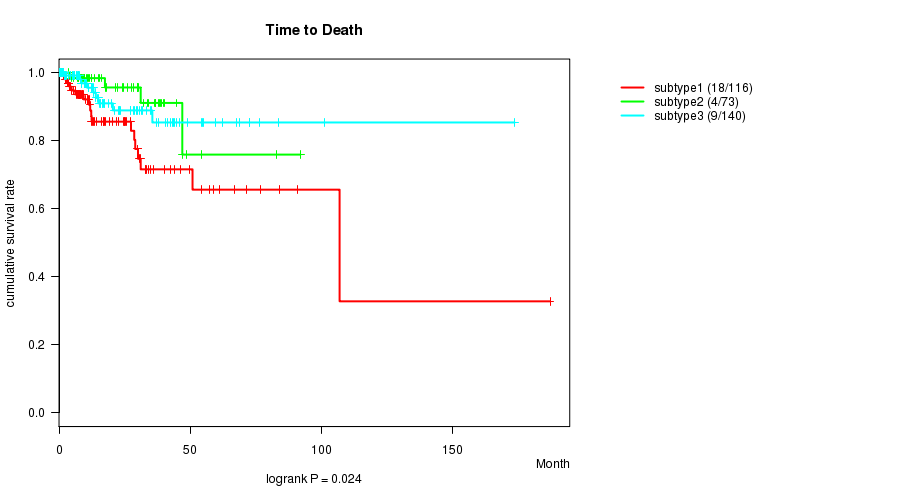
P value = 0.0445 (ANOVA), Q value = 0.76
Table S18. Clustering Approach #4: 'METHLYATION CNMF' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
| ALL | 333 | 63.6 (11.3) |
| subtype1 | 118 | 65.7 (10.6) |
| subtype2 | 73 | 62.9 (12.9) |
| subtype3 | 142 | 62.3 (10.8) |
Figure S14. Get High-res Image Clustering Approach #4: 'METHLYATION CNMF' versus Clinical Feature #2: 'AGE'
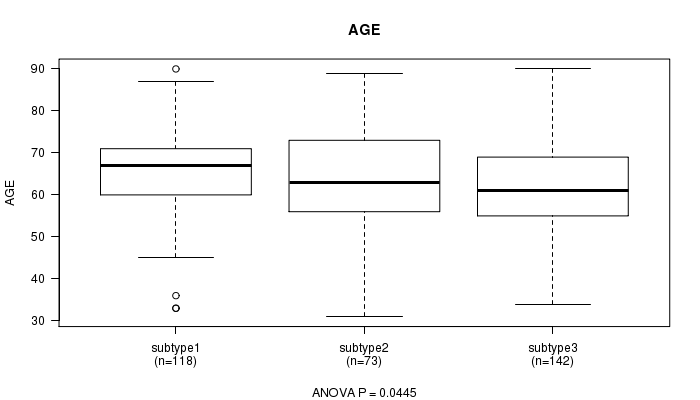
P value = 2.17e-18 (Chi-square test), Q value = 8e-17
Table S19. Clustering Approach #4: 'METHLYATION CNMF' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 3) | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|---|---|---|
| ALL | 245 | 6 | 1 | 3 | 17 | 62 |
| subtype1 | 54 | 0 | 0 | 1 | 12 | 52 |
| subtype2 | 57 | 2 | 1 | 0 | 3 | 10 |
| subtype3 | 134 | 4 | 0 | 2 | 2 | 0 |
Figure S15. Get High-res Image Clustering Approach #4: 'METHLYATION CNMF' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
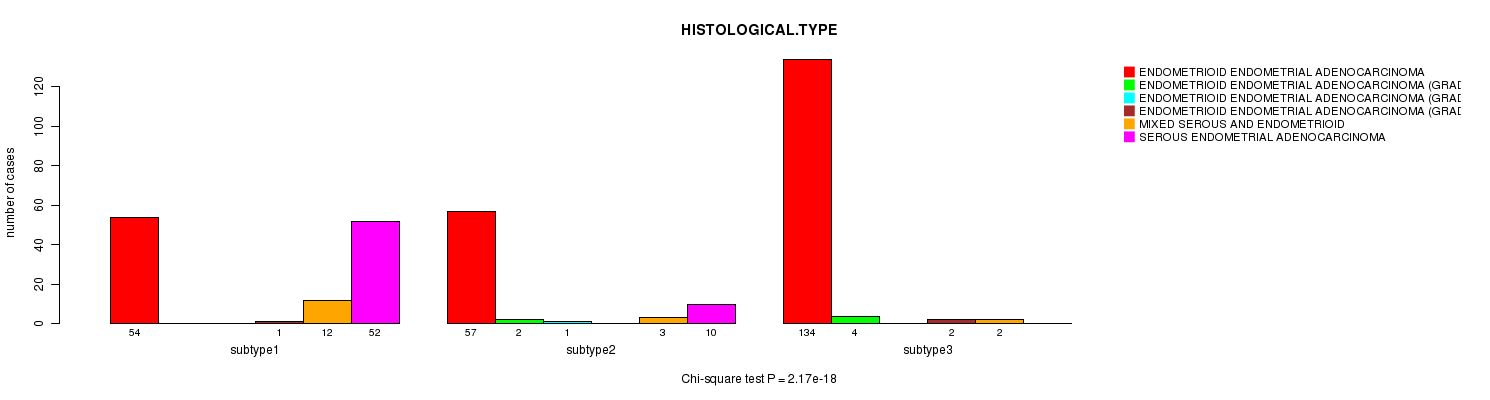
P value = 0.206 (Fisher's exact test), Q value = 1
Table S20. Clustering Approach #4: 'METHLYATION CNMF' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 81 | 253 |
| subtype1 | 32 | 87 |
| subtype2 | 12 | 61 |
| subtype3 | 37 | 105 |
Figure S16. Get High-res Image Clustering Approach #4: 'METHLYATION CNMF' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
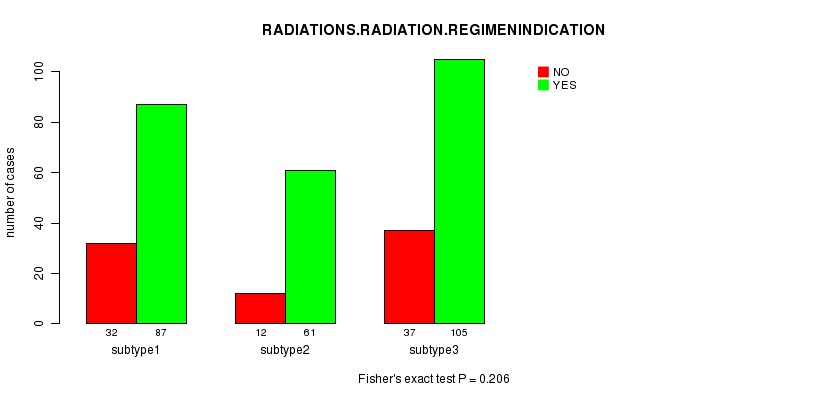
Table S21. Get Full Table Description of clustering approach #5: 'RPPA CNMF subtypes'
| Cluster Labels | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| Number of samples | 41 | 38 | 41 | 16 | 38 | 26 |
P value = 0.778 (logrank test), Q value = 1
Table S22. Clustering Approach #5: 'RPPA CNMF subtypes' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 200 | 13 | 0.0 - 173.6 (21.7) |
| subtype1 | 41 | 4 | 0.6 - 106.9 (21.0) |
| subtype2 | 38 | 2 | 1.3 - 133.2 (24.7) |
| subtype3 | 41 | 2 | 1.8 - 173.6 (22.6) |
| subtype4 | 16 | 2 | 1.4 - 82.7 (26.6) |
| subtype5 | 38 | 1 | 0.0 - 101.1 (12.4) |
| subtype6 | 26 | 2 | 0.7 - 66.9 (20.8) |
Figure S17. Get High-res Image Clustering Approach #5: 'RPPA CNMF subtypes' versus Clinical Feature #1: 'Time to Death'
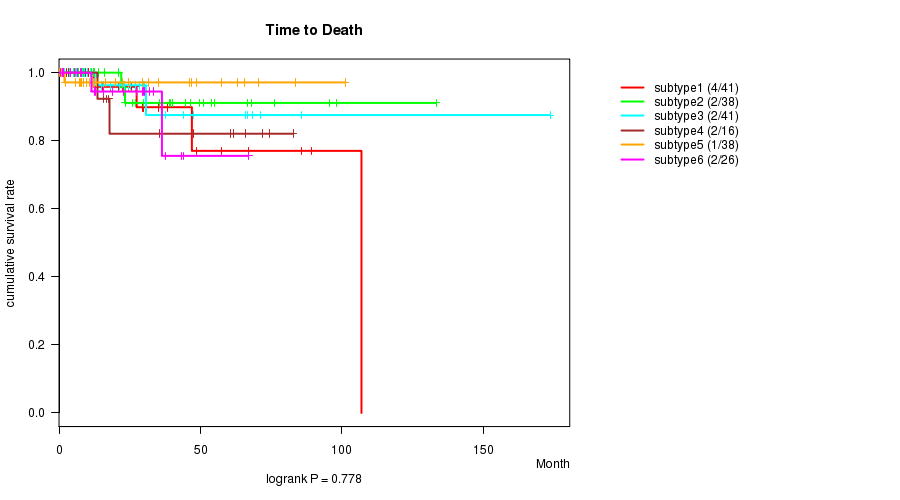
P value = 0.343 (ANOVA), Q value = 1
Table S23. Clustering Approach #5: 'RPPA CNMF subtypes' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
| ALL | 200 | 62.7 (11.1) |
| subtype1 | 41 | 62.9 (12.2) |
| subtype2 | 38 | 63.4 (10.6) |
| subtype3 | 41 | 61.9 (10.8) |
| subtype4 | 16 | 68.1 (8.2) |
| subtype5 | 38 | 60.6 (9.6) |
| subtype6 | 26 | 62.6 (13.3) |
Figure S18. Get High-res Image Clustering Approach #5: 'RPPA CNMF subtypes' versus Clinical Feature #2: 'AGE'
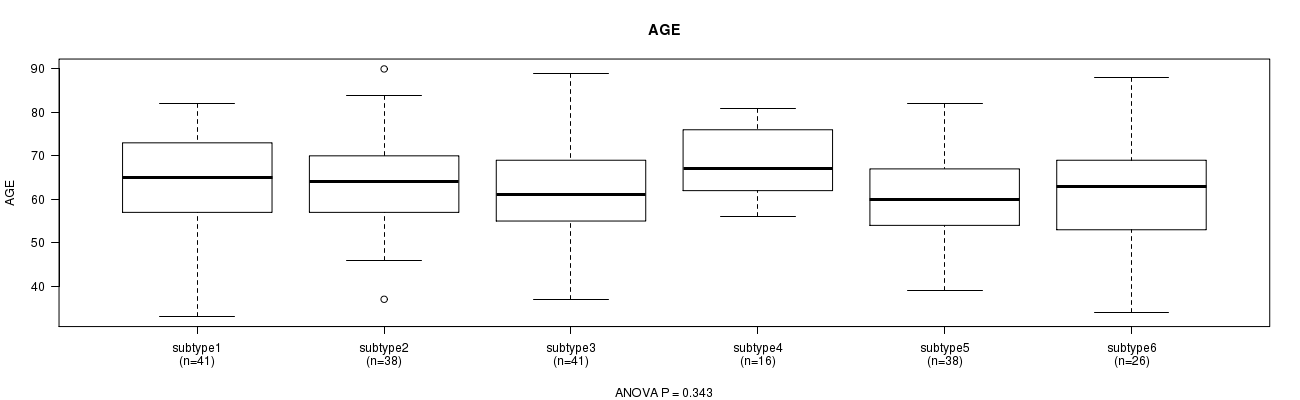
P value = 3.91e-06 (Chi-square test), Q value = 0.00012
Table S24. Clustering Approach #5: 'RPPA CNMF subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1 OR 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 3) | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|---|---|---|
| ALL | 164 | 3 | 3 | 4 | 3 | 23 |
| subtype1 | 33 | 0 | 0 | 1 | 1 | 6 |
| subtype2 | 26 | 0 | 0 | 1 | 0 | 11 |
| subtype3 | 40 | 0 | 1 | 0 | 0 | 0 |
| subtype4 | 12 | 2 | 0 | 2 | 0 | 0 |
| subtype5 | 36 | 0 | 2 | 0 | 0 | 0 |
| subtype6 | 17 | 1 | 0 | 0 | 2 | 6 |
Figure S19. Get High-res Image Clustering Approach #5: 'RPPA CNMF subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
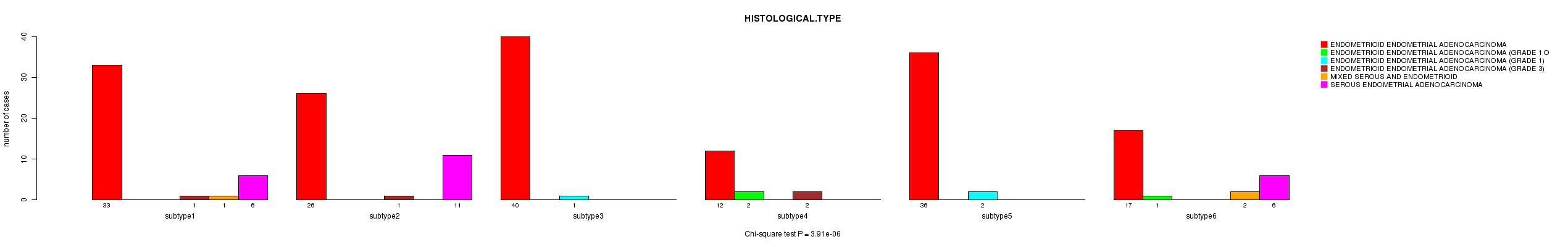
P value = 0.327 (Chi-square test), Q value = 1
Table S25. Clustering Approach #5: 'RPPA CNMF subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 80 | 120 |
| subtype1 | 20 | 21 |
| subtype2 | 18 | 20 |
| subtype3 | 13 | 28 |
| subtype4 | 4 | 12 |
| subtype5 | 13 | 25 |
| subtype6 | 12 | 14 |
Figure S20. Get High-res Image Clustering Approach #5: 'RPPA CNMF subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
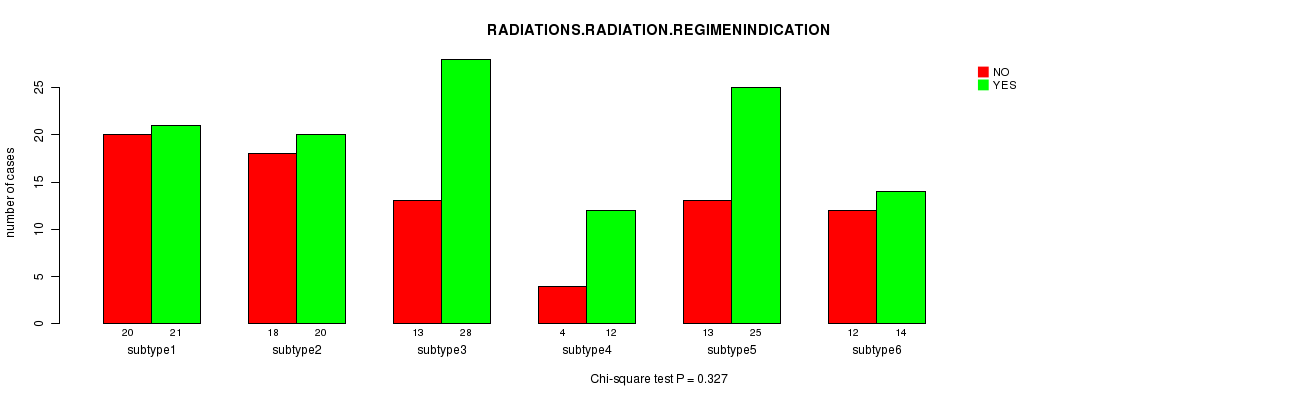
Table S26. Get Full Table Description of clustering approach #6: 'RPPA cHierClus subtypes'
| Cluster Labels | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| Number of samples | 7 | 33 | 39 | 39 | 56 | 26 |
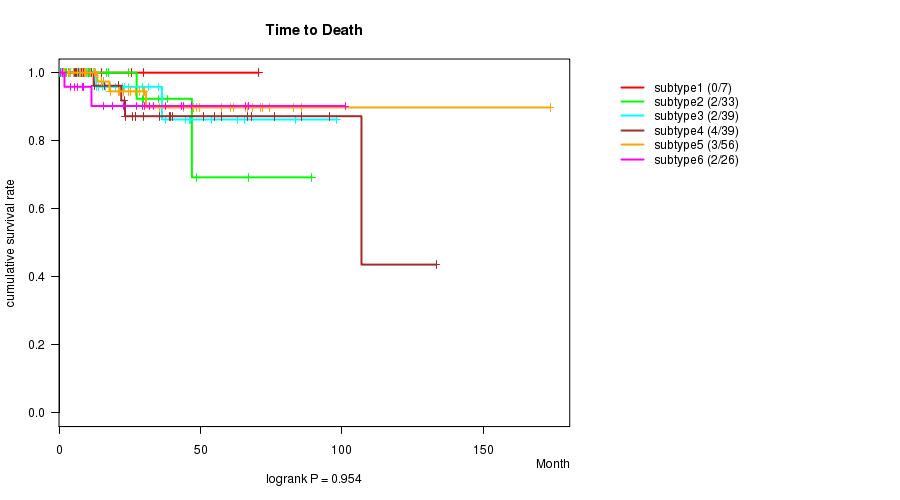
P value = 0.954 (logrank test), Q value = 1
Table S27. Clustering Approach #6: 'RPPA cHierClus subtypes' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 200 | 13 | 0.0 - 173.6 (21.7) |
| subtype1 | 7 | 0 | 9.2 - 70.4 (15.1) |
| subtype2 | 33 | 2 | 0.0 - 89.3 (12.2) |
| subtype3 | 39 | 2 | 0.7 - 98.2 (16.3) |
| subtype4 | 39 | 4 | 1.3 - 133.2 (23.3) |
| subtype5 | 56 | 3 | 1.4 - 173.6 (23.7) |
| subtype6 | 26 | 2 | 0.7 - 101.1 (22.7) |
Figure S21. Get High-res Image Clustering Approach #6: 'RPPA cHierClus subtypes' versus Clinical Feature #1: 'Time to Death'
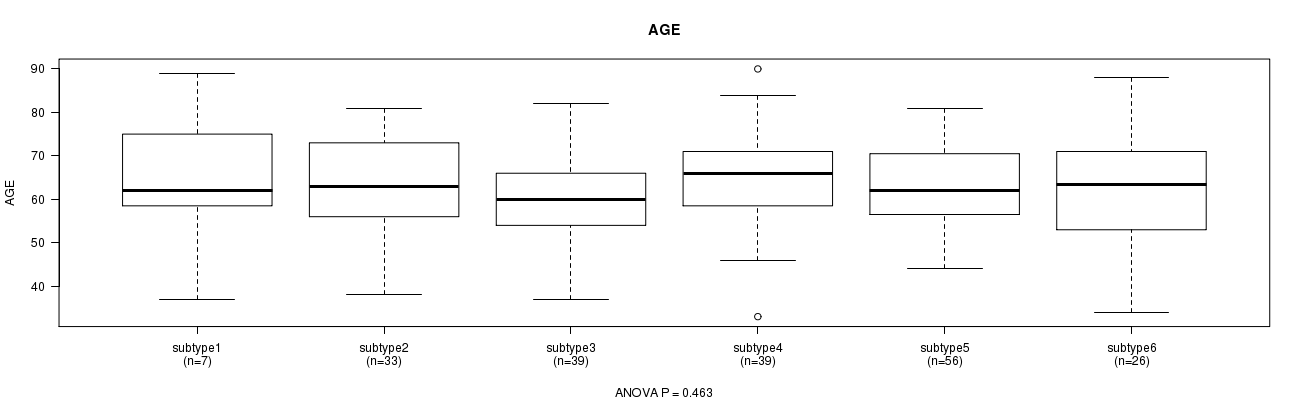
P value = 0.463 (ANOVA), Q value = 1
Table S28. Clustering Approach #6: 'RPPA cHierClus subtypes' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
| ALL | 200 | 62.7 (11.1) |
| subtype1 | 7 | 65.0 (17.0) |
| subtype2 | 33 | 62.5 (11.5) |
| subtype3 | 39 | 59.8 (9.3) |
| subtype4 | 39 | 65.0 (11.3) |
| subtype5 | 56 | 63.1 (9.8) |
| subtype6 | 26 | 62.5 (13.2) |
Figure S22. Get High-res Image Clustering Approach #6: 'RPPA cHierClus subtypes' versus Clinical Feature #2: 'AGE'
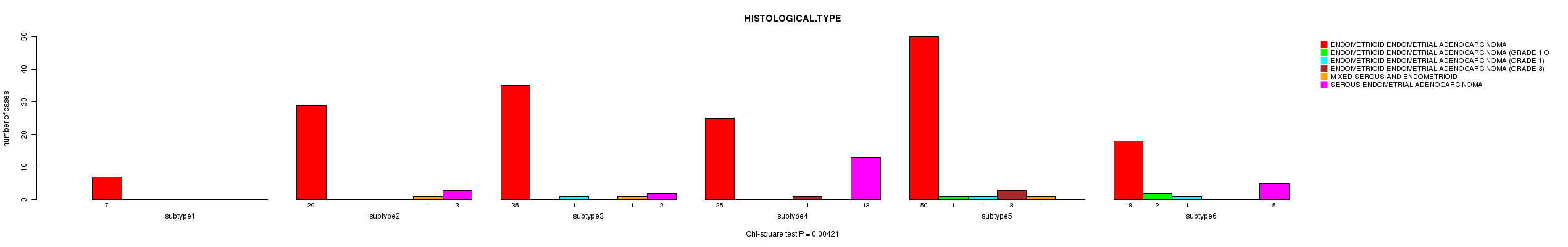
P value = 0.00421 (Chi-square test), Q value = 0.097
Table S29. Clustering Approach #6: 'RPPA cHierClus subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1 OR 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 3) | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|---|---|---|
| ALL | 164 | 3 | 3 | 4 | 3 | 23 |
| subtype1 | 7 | 0 | 0 | 0 | 0 | 0 |
| subtype2 | 29 | 0 | 0 | 0 | 1 | 3 |
| subtype3 | 35 | 0 | 1 | 0 | 1 | 2 |
| subtype4 | 25 | 0 | 0 | 1 | 0 | 13 |
| subtype5 | 50 | 1 | 1 | 3 | 1 | 0 |
| subtype6 | 18 | 2 | 1 | 0 | 0 | 5 |
Figure S23. Get High-res Image Clustering Approach #6: 'RPPA cHierClus subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
P value = 0.584 (Chi-square test), Q value = 1
Table S30. Clustering Approach #6: 'RPPA cHierClus subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 80 | 120 |
| subtype1 | 2 | 5 |
| subtype2 | 10 | 23 |
| subtype3 | 15 | 24 |
| subtype4 | 19 | 20 |
| subtype5 | 25 | 31 |
| subtype6 | 9 | 17 |
Figure S24. Get High-res Image Clustering Approach #6: 'RPPA cHierClus subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
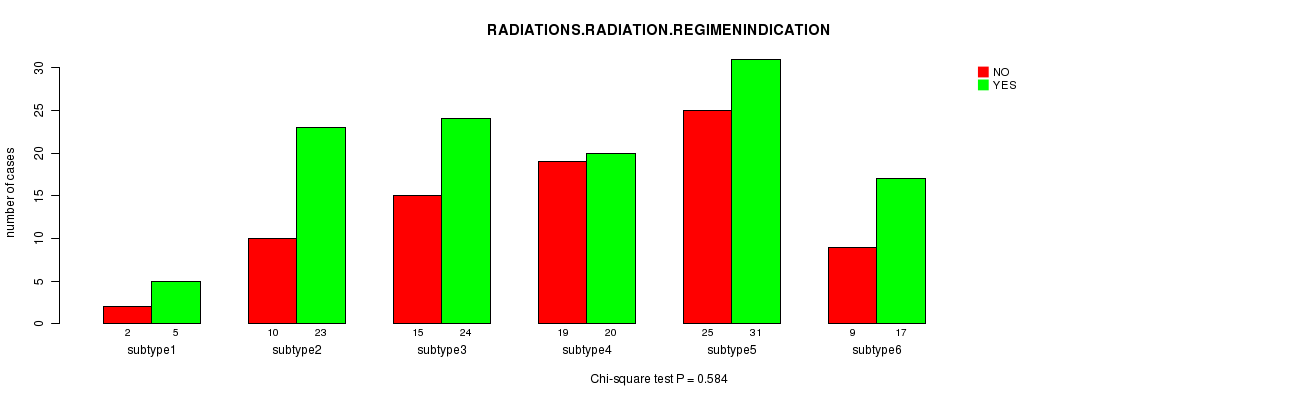
Table S31. Get Full Table Description of clustering approach #7: 'RNAseq CNMF subtypes'
| Cluster Labels | 1 | 2 | 3 |
|---|---|---|---|
| Number of samples | 142 | 113 | 114 |
P value = 0.00308 (logrank test), Q value = 0.074
Table S32. Clustering Approach #7: 'RNAseq CNMF subtypes' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 367 | 32 | 0.0 - 187.1 (17.8) |
| subtype1 | 141 | 21 | 0.1 - 187.1 (15.3) |
| subtype2 | 112 | 3 | 0.3 - 101.1 (17.8) |
| subtype3 | 114 | 8 | 0.0 - 173.6 (22.7) |
Figure S25. Get High-res Image Clustering Approach #7: 'RNAseq CNMF subtypes' versus Clinical Feature #1: 'Time to Death'
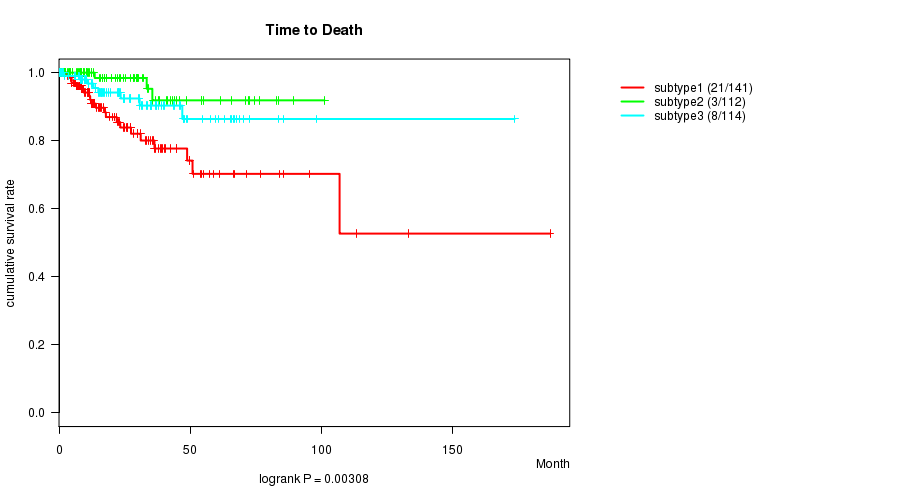
P value = 1.1e-06 (ANOVA), Q value = 3.5e-05
Table S33. Clustering Approach #7: 'RNAseq CNMF subtypes' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
| ALL | 369 | 63.2 (11.0) |
| subtype1 | 142 | 66.3 (10.0) |
| subtype2 | 113 | 63.4 (10.8) |
| subtype3 | 114 | 59.2 (11.2) |
Figure S26. Get High-res Image Clustering Approach #7: 'RNAseq CNMF subtypes' versus Clinical Feature #2: 'AGE'
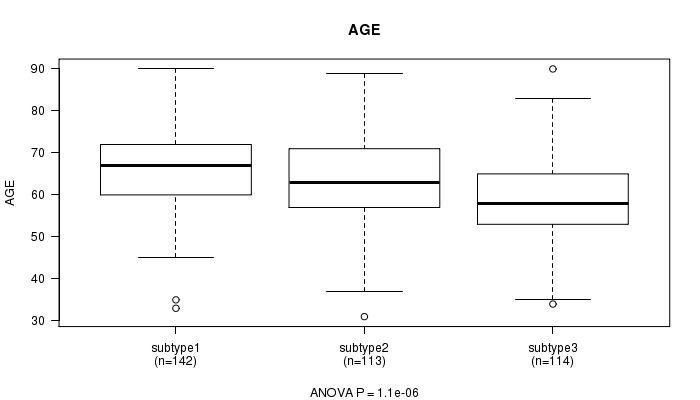
P value = 2.35e-23 (Chi-square test), Q value = 8.9e-22
Table S34. Clustering Approach #7: 'RNAseq CNMF subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1 OR 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 3) | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|---|---|---|---|
| ALL | 283 | 3 | 7 | 2 | 7 | 10 | 57 |
| subtype1 | 73 | 0 | 0 | 0 | 3 | 9 | 57 |
| subtype2 | 106 | 2 | 4 | 0 | 0 | 1 | 0 |
| subtype3 | 104 | 1 | 3 | 2 | 4 | 0 | 0 |
Figure S27. Get High-res Image Clustering Approach #7: 'RNAseq CNMF subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
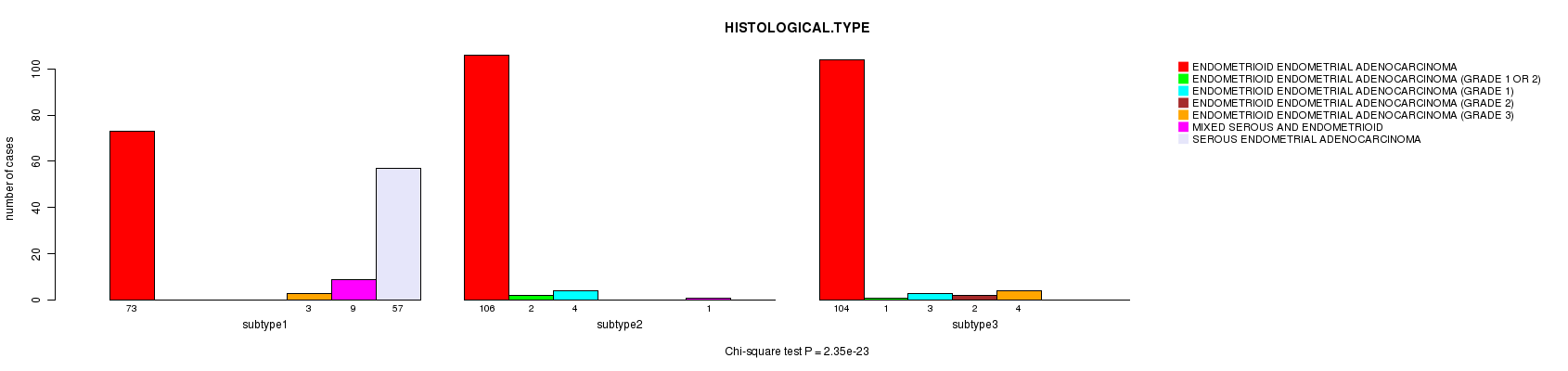
P value = 0.0553 (Fisher's exact test), Q value = 0.88
Table S35. Clustering Approach #7: 'RNAseq CNMF subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 123 | 246 |
| subtype1 | 55 | 87 |
| subtype2 | 28 | 85 |
| subtype3 | 40 | 74 |
Figure S28. Get High-res Image Clustering Approach #7: 'RNAseq CNMF subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
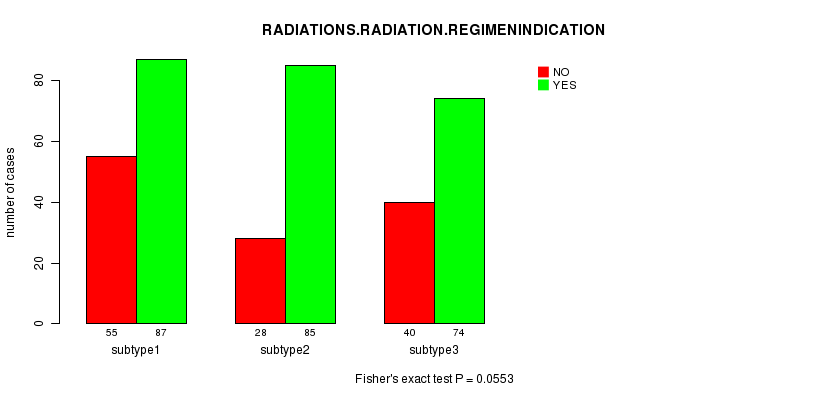
Table S36. Get Full Table Description of clustering approach #8: 'RNAseq cHierClus subtypes'
| Cluster Labels | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| Number of samples | 65 | 98 | 65 | 141 |
P value = 0.0985 (logrank test), Q value = 1
Table S37. Clustering Approach #8: 'RNAseq cHierClus subtypes' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 367 | 32 | 0.0 - 187.1 (17.8) |
| subtype1 | 64 | 6 | 0.0 - 173.6 (17.5) |
| subtype2 | 98 | 2 | 0.3 - 101.1 (17.0) |
| subtype3 | 65 | 8 | 0.3 - 106.9 (25.0) |
| subtype4 | 140 | 16 | 0.1 - 187.1 (16.5) |
Figure S29. Get High-res Image Clustering Approach #8: 'RNAseq cHierClus subtypes' versus Clinical Feature #1: 'Time to Death'
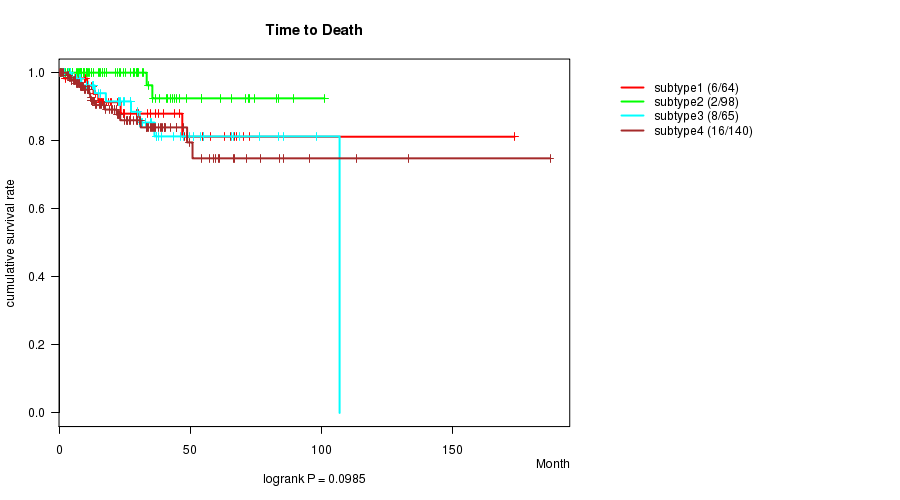
P value = 8.03e-07 (ANOVA), Q value = 2.7e-05
Table S38. Clustering Approach #8: 'RNAseq cHierClus subtypes' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
| ALL | 369 | 63.2 (11.0) |
| subtype1 | 65 | 58.3 (10.4) |
| subtype2 | 98 | 62.8 (10.9) |
| subtype3 | 65 | 61.1 (11.5) |
| subtype4 | 141 | 66.8 (10.0) |
Figure S30. Get High-res Image Clustering Approach #8: 'RNAseq cHierClus subtypes' versus Clinical Feature #2: 'AGE'
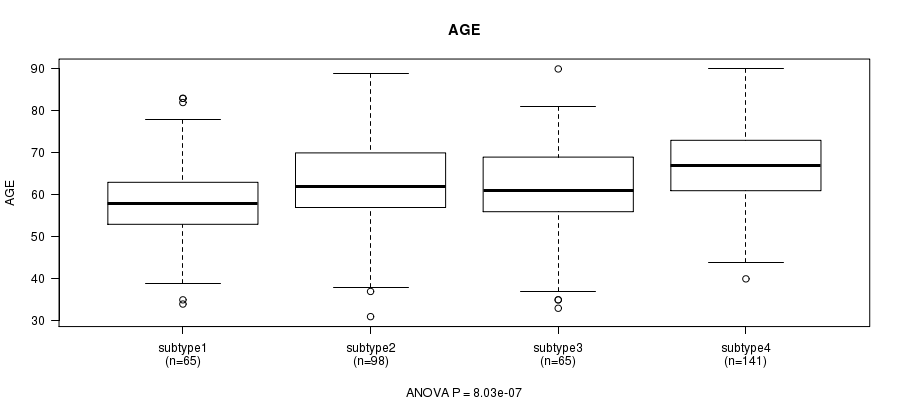
P value = 2.96e-17 (Chi-square test), Q value = 1e-15
Table S39. Clustering Approach #8: 'RNAseq cHierClus subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1 OR 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 3) | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|---|---|---|---|
| ALL | 283 | 3 | 7 | 2 | 7 | 10 | 57 |
| subtype1 | 59 | 1 | 1 | 2 | 2 | 0 | 0 |
| subtype2 | 91 | 2 | 4 | 0 | 0 | 1 | 0 |
| subtype3 | 57 | 0 | 2 | 0 | 1 | 2 | 3 |
| subtype4 | 76 | 0 | 0 | 0 | 4 | 7 | 54 |
Figure S31. Get High-res Image Clustering Approach #8: 'RNAseq cHierClus subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
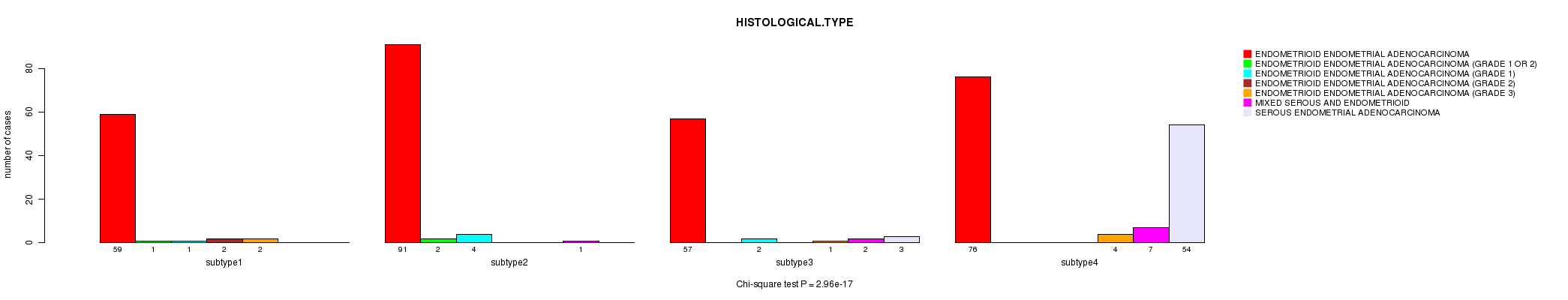
P value = 0.00715 (Fisher's exact test), Q value = 0.15
Table S40. Clustering Approach #8: 'RNAseq cHierClus subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 123 | 246 |
| subtype1 | 18 | 47 |
| subtype2 | 25 | 73 |
| subtype3 | 33 | 32 |
| subtype4 | 47 | 94 |
Figure S32. Get High-res Image Clustering Approach #8: 'RNAseq cHierClus subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
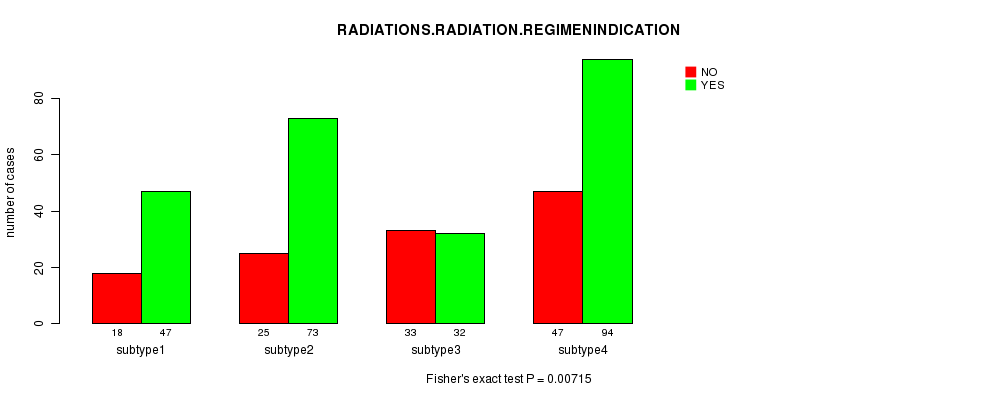
Table S41. Get Full Table Description of clustering approach #9: 'MIRseq CNMF subtypes'
| Cluster Labels | 1 | 2 | 3 |
|---|---|---|---|
| Number of samples | 159 | 146 | 131 |
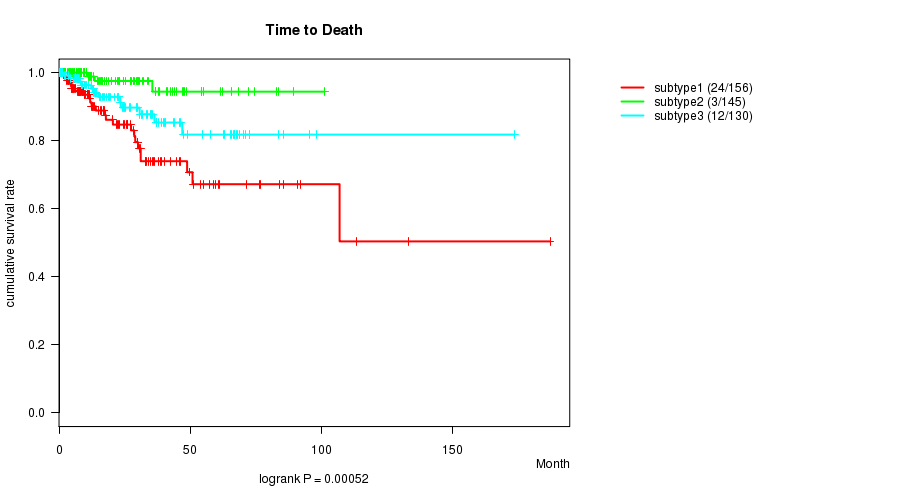
P value = 0.00052 (logrank test), Q value = 0.015
Table S42. Clustering Approach #9: 'MIRseq CNMF subtypes' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 431 | 39 | 0.0 - 187.1 (15.7) |
| subtype1 | 156 | 24 | 0.0 - 187.1 (12.4) |
| subtype2 | 145 | 3 | 0.1 - 101.1 (15.2) |
| subtype3 | 130 | 12 | 0.2 - 173.6 (19.4) |
Figure S33. Get High-res Image Clustering Approach #9: 'MIRseq CNMF subtypes' versus Clinical Feature #1: 'Time to Death'
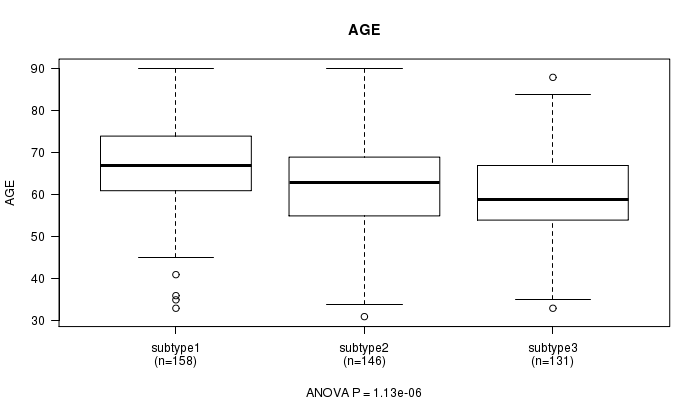
P value = 1.13e-06 (ANOVA), Q value = 3.5e-05
Table S43. Clustering Approach #9: 'MIRseq CNMF subtypes' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
| ALL | 435 | 63.4 (11.2) |
| subtype1 | 158 | 66.8 (10.5) |
| subtype2 | 146 | 62.5 (11.2) |
| subtype3 | 131 | 60.2 (11.0) |
Figure S34. Get High-res Image Clustering Approach #9: 'MIRseq CNMF subtypes' versus Clinical Feature #2: 'AGE'
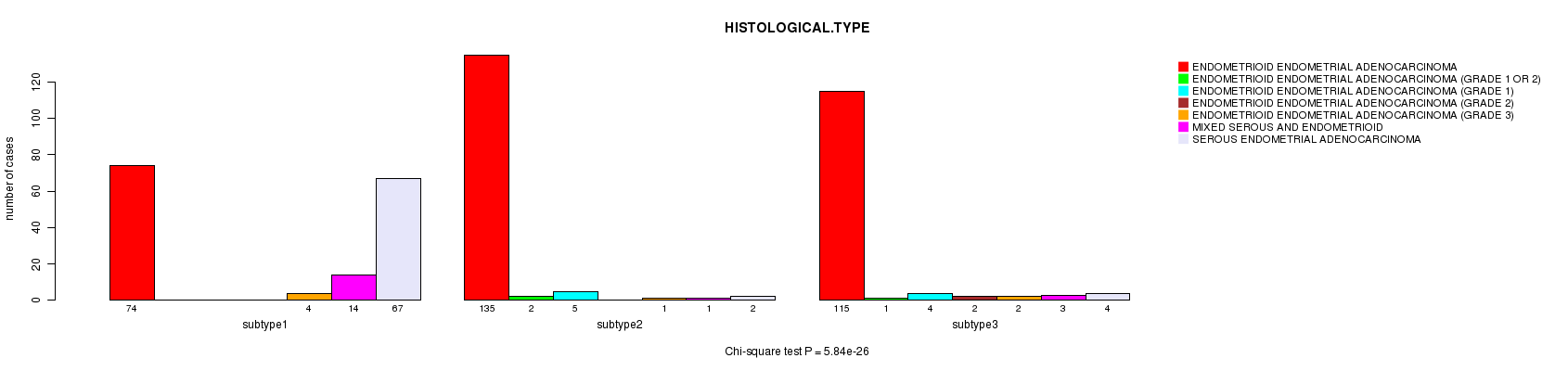
P value = 5.84e-26 (Chi-square test), Q value = 2.3e-24
Table S44. Clustering Approach #9: 'MIRseq CNMF subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1 OR 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 3) | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|---|---|---|---|
| ALL | 324 | 3 | 9 | 2 | 7 | 18 | 73 |
| subtype1 | 74 | 0 | 0 | 0 | 4 | 14 | 67 |
| subtype2 | 135 | 2 | 5 | 0 | 1 | 1 | 2 |
| subtype3 | 115 | 1 | 4 | 2 | 2 | 3 | 4 |
Figure S35. Get High-res Image Clustering Approach #9: 'MIRseq CNMF subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
P value = 0.406 (Fisher's exact test), Q value = 1
Table S45. Clustering Approach #9: 'MIRseq CNMF subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 127 | 309 |
| subtype1 | 44 | 115 |
| subtype2 | 39 | 107 |
| subtype3 | 44 | 87 |
Figure S36. Get High-res Image Clustering Approach #9: 'MIRseq CNMF subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
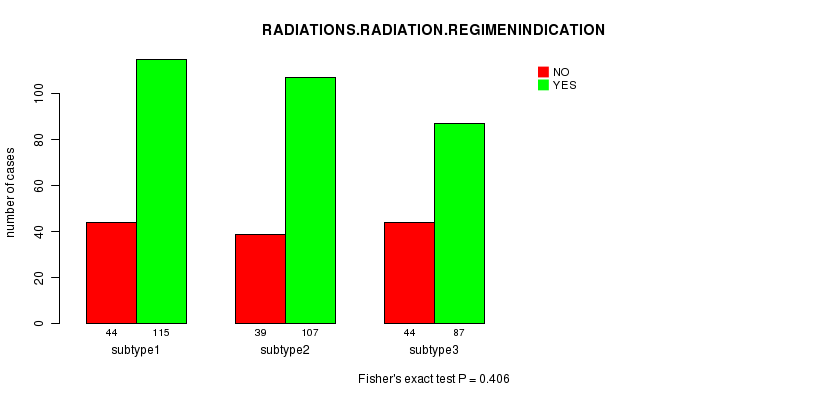
Table S46. Get Full Table Description of clustering approach #10: 'MIRseq cHierClus subtypes'
| Cluster Labels | 1 | 2 | 3 |
|---|---|---|---|
| Number of samples | 24 | 207 | 205 |
P value = 0.00112 (logrank test), Q value = 0.03
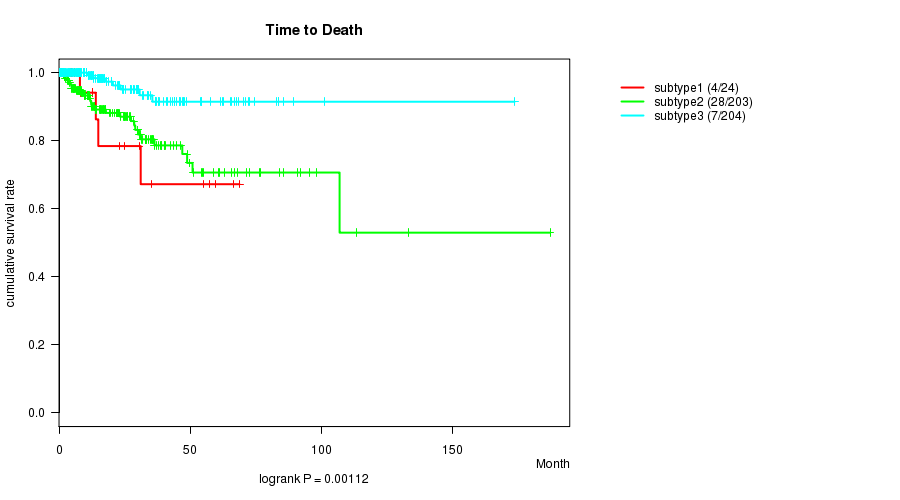
Table S47. Clustering Approach #10: 'MIRseq cHierClus subtypes' versus Clinical Feature #1: 'Time to Death'
| nPatients | nDeath | Duration Range (Median), Month | |
|---|---|---|---|
| ALL | 431 | 39 | 0.0 - 187.1 (15.7) |
| subtype1 | 24 | 4 | 0.5 - 68.7 (13.4) |
| subtype2 | 203 | 28 | 0.0 - 187.1 (14.0) |
| subtype3 | 204 | 7 | 0.0 - 173.6 (16.6) |
Figure S37. Get High-res Image Clustering Approach #10: 'MIRseq cHierClus subtypes' versus Clinical Feature #1: 'Time to Death'
P value = 0.00244 (ANOVA), Q value = 0.061
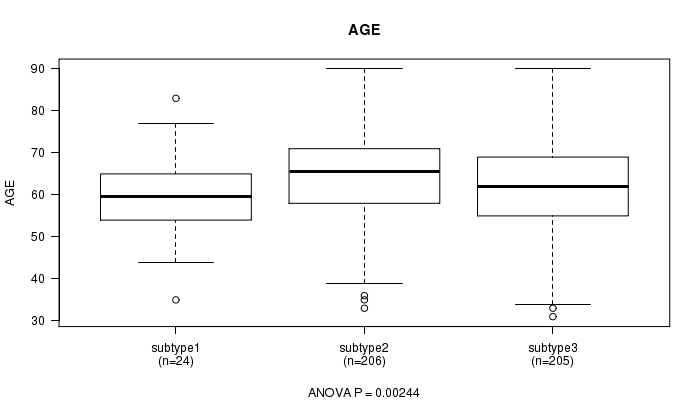
Table S48. Clustering Approach #10: 'MIRseq cHierClus subtypes' versus Clinical Feature #2: 'AGE'
| nPatients | Mean (Std.Dev) | |
|---|---|---|
| ALL | 435 | 63.4 (11.2) |
| subtype1 | 24 | 59.6 (11.4) |
| subtype2 | 206 | 65.3 (10.7) |
| subtype3 | 205 | 61.9 (11.4) |
Figure S38. Get High-res Image Clustering Approach #10: 'MIRseq cHierClus subtypes' versus Clinical Feature #2: 'AGE'
P value = 2.28e-18 (Chi-square test), Q value = 8.2e-17
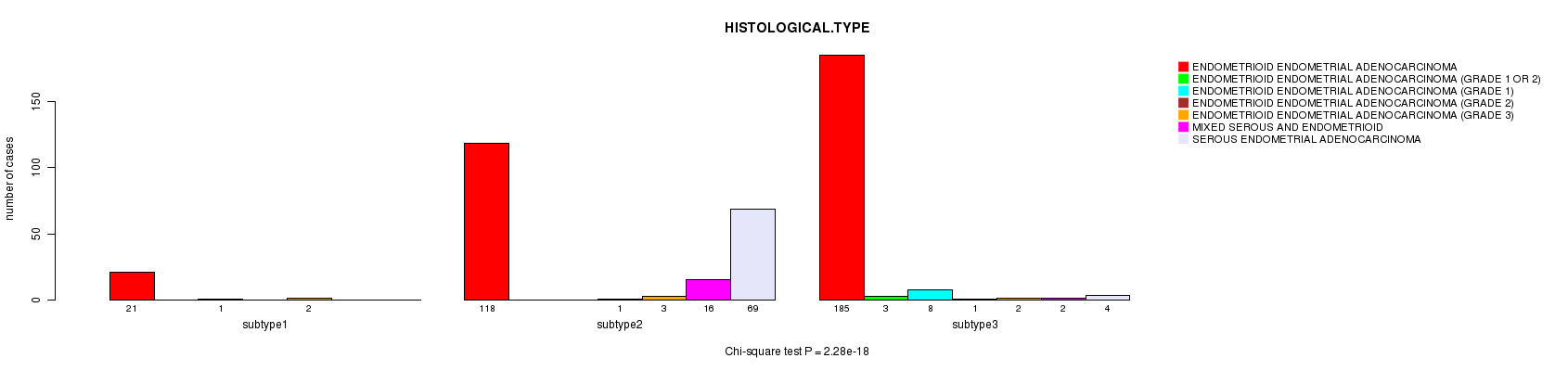
Table S49. Clustering Approach #10: 'MIRseq cHierClus subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
| nPatients | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1 OR 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 1) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 2) | ENDOMETRIOID ENDOMETRIAL ADENOCARCINOMA (GRADE 3) | MIXED SEROUS AND ENDOMETRIOID | SEROUS ENDOMETRIAL ADENOCARCINOMA |
|---|---|---|---|---|---|---|---|
| ALL | 324 | 3 | 9 | 2 | 7 | 18 | 73 |
| subtype1 | 21 | 0 | 1 | 0 | 2 | 0 | 0 |
| subtype2 | 118 | 0 | 0 | 1 | 3 | 16 | 69 |
| subtype3 | 185 | 3 | 8 | 1 | 2 | 2 | 4 |
Figure S39. Get High-res Image Clustering Approach #10: 'MIRseq cHierClus subtypes' versus Clinical Feature #3: 'HISTOLOGICAL.TYPE'
P value = 0.207 (Fisher's exact test), Q value = 1
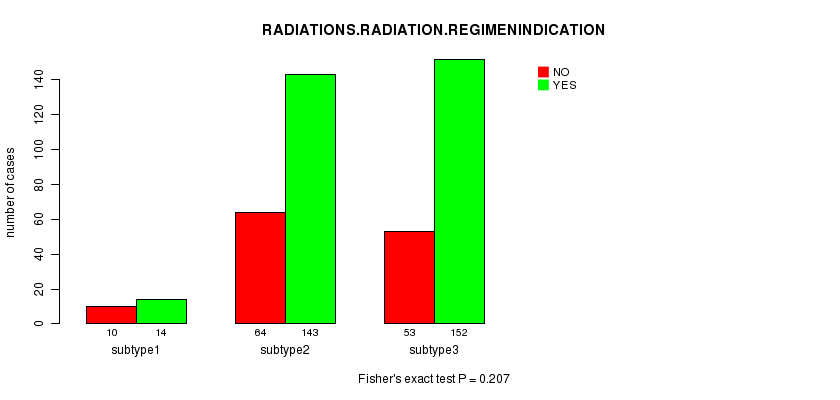
Table S50. Clustering Approach #10: 'MIRseq cHierClus subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
| nPatients | NO | YES |
|---|---|---|
| ALL | 127 | 309 |
| subtype1 | 10 | 14 |
| subtype2 | 64 | 143 |
| subtype3 | 53 | 152 |
Figure S40. Get High-res Image Clustering Approach #10: 'MIRseq cHierClus subtypes' versus Clinical Feature #4: 'RADIATIONS.RADIATION.REGIMENINDICATION'
-
Cluster data file = UCEC-TP.mergedcluster.txt
-
Clinical data file = UCEC-TP.clin.merged.picked.txt
-
Number of patients = 451
-
Number of clustering approaches = 10
-
Number of selected clinical features = 4
-
Exclude small clusters that include fewer than K patients, K = 3
consensus non-negative matrix factorization clustering approach (Brunet et al. 2004)
Resampling-based clustering method (Monti et al. 2003)
For survival clinical features, the Kaplan-Meier survival curves of tumors with and without gene mutations were plotted and the statistical significance P values were estimated by logrank test (Bland and Altman 2004) using the 'survdiff' function in R
For continuous numerical clinical features, one-way analysis of variance (Howell 2002) was applied to compare the clinical values between tumor subtypes using 'anova' function in R
For multi-class clinical features (nominal or ordinal), Chi-square tests (Greenwood and Nikulin 1996) were used to estimate the P values using the 'chisq.test' function in R
For binary clinical features, two-tailed Fisher's exact tests (Fisher 1922) were used to estimate the P values using the 'fisher.test' function in R
For multiple hypothesis correction, Q value is the False Discovery Rate (FDR) analogue of the P value (Benjamini and Hochberg 1995), defined as the minimum FDR at which the test may be called significant. We used the 'Benjamini and Hochberg' method of 'p.adjust' function in R to convert P values into Q values.
This is an experimental feature. The full results of the analysis summarized in this report can be downloaded from the TCGA Data Coordination Center.