This pipeline calculates clusters based on a consensus non-negative matrix factorization (NMF) clustering method ##REF##1,##REF##3. This pipeline has the following features:
-
Convert input data set to a non-negitive matrix by column rank normalization.
-
Classify samples into consensus clusters.
-
Determine differentially expressed marker genes for each subtype.
The most robust consensus NMF clustering of 122 samples using the 1500 most variable genes was identified for k = 3 clusters. We computed the clustering for k = 2 to k = 8 and used the cophenetic correlation coefficient to determine the best solution.
Figure 1. Get High-res Image Samples were separated into 3 clusters. Shown are 122 samples and 900 marker genes. The color bar of the row indicates the marker genes for the corresponding cluster.
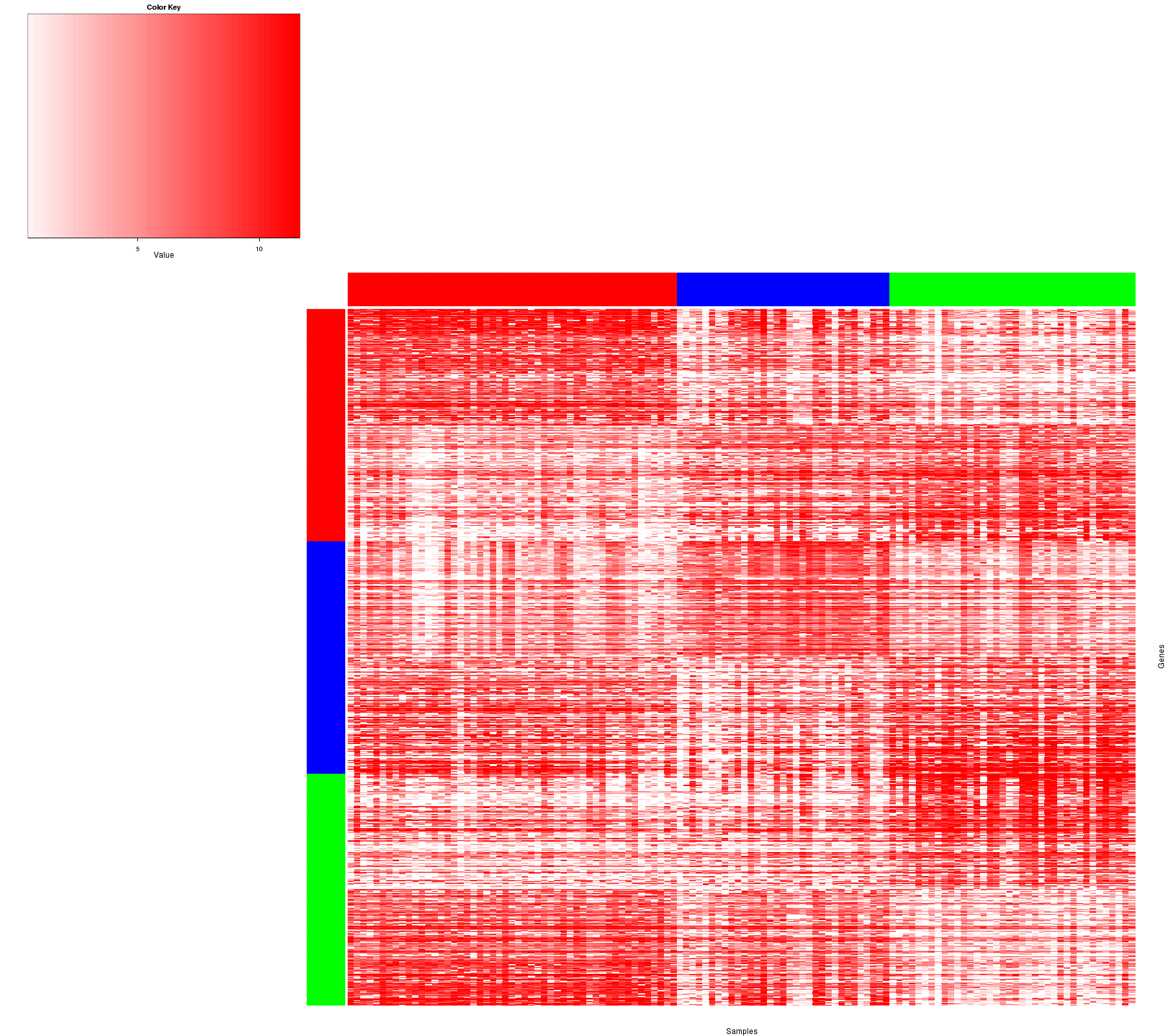
Figure 2. Get High-res Image Heatmap with a standard hierarchical clustering for 122 samples and the 1500 most variable genes.
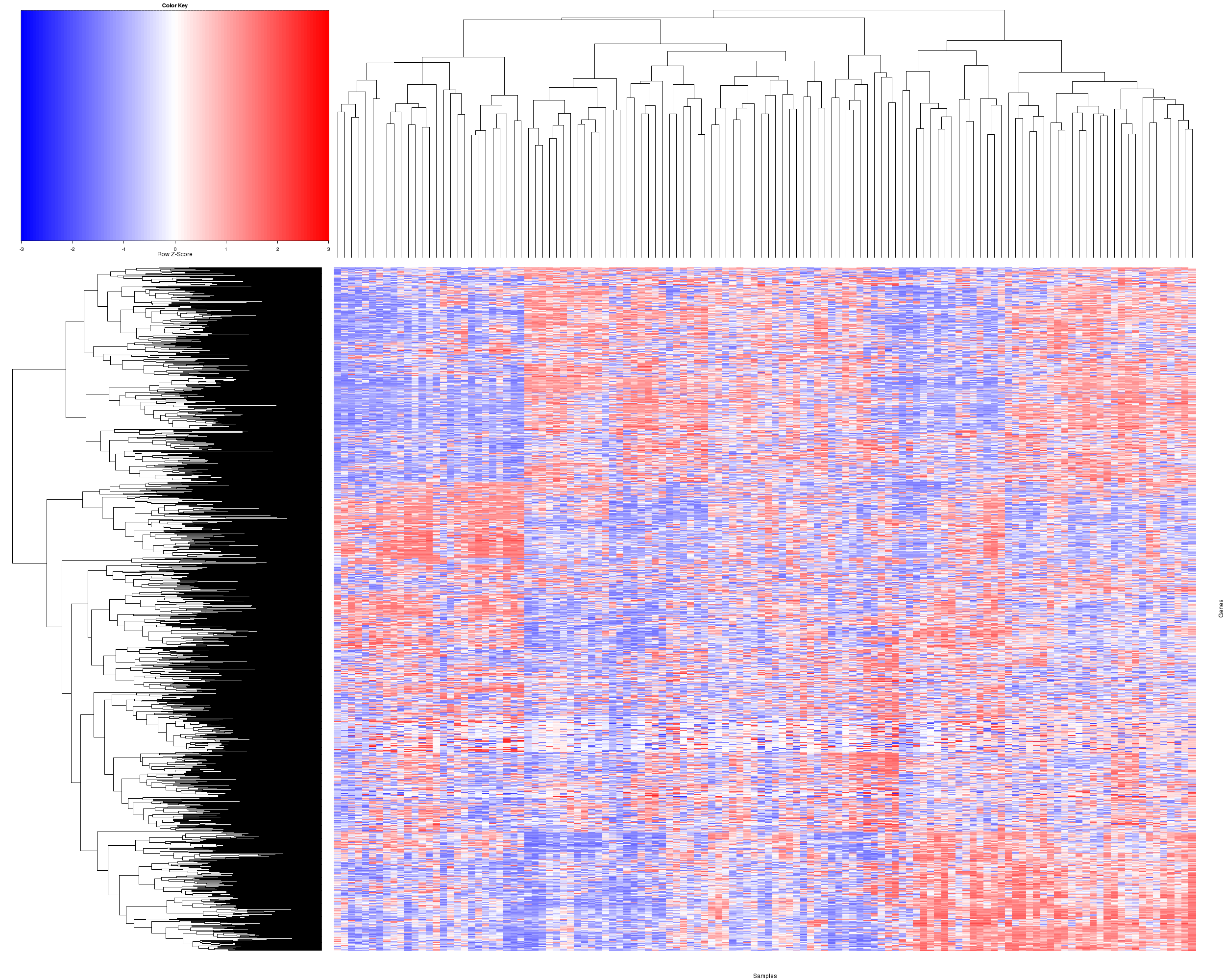
Figure 3. Get High-res Image The consensus matrix after clustering shows 3 clusters with limited overlap between clusters.
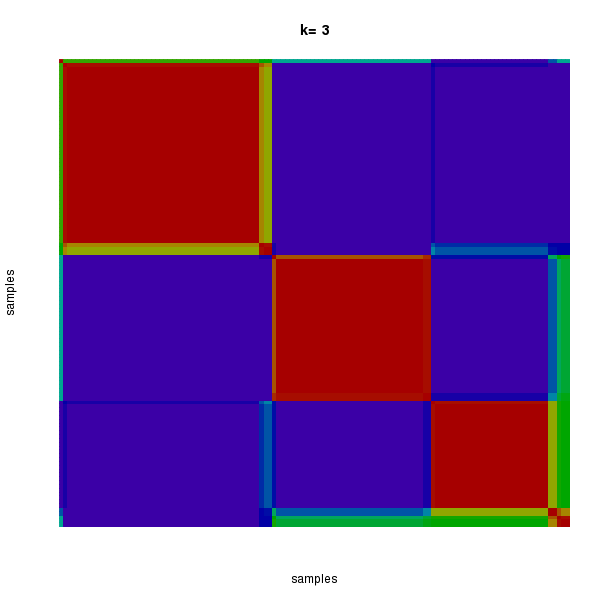
Figure 4. Get High-res Image The correlation matrix also shows 3 clusters.
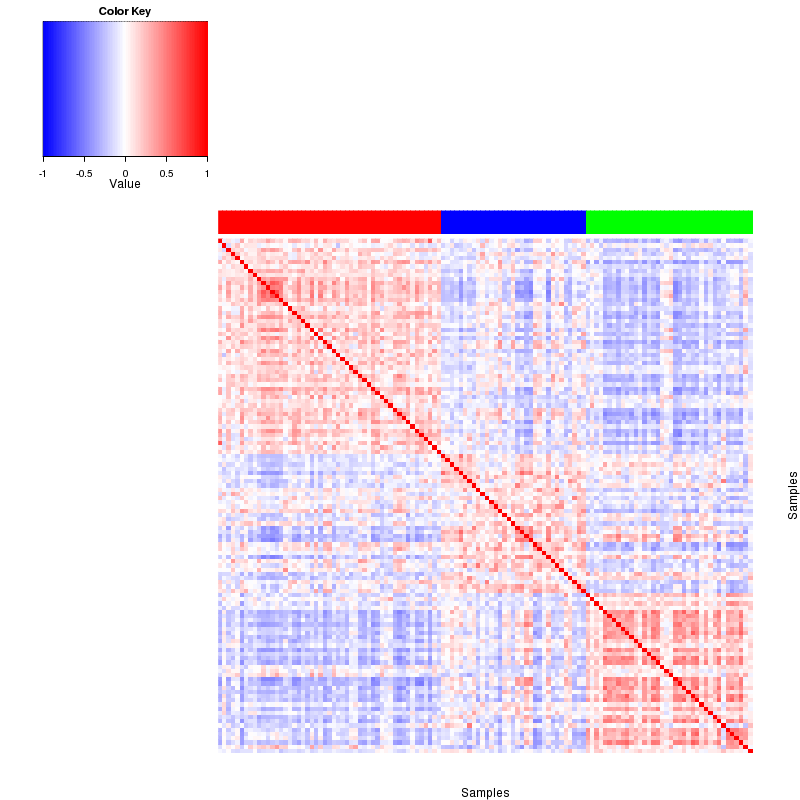
Figure 5. Get High-res Image The silhouette width was calculated for each sample and each value of k. The left panel shows the average silhouette width across all samples for each tested k (left panel). The right panels shows assignments of clusters to samples and the silhouette width of each sample for the most robust clustering.
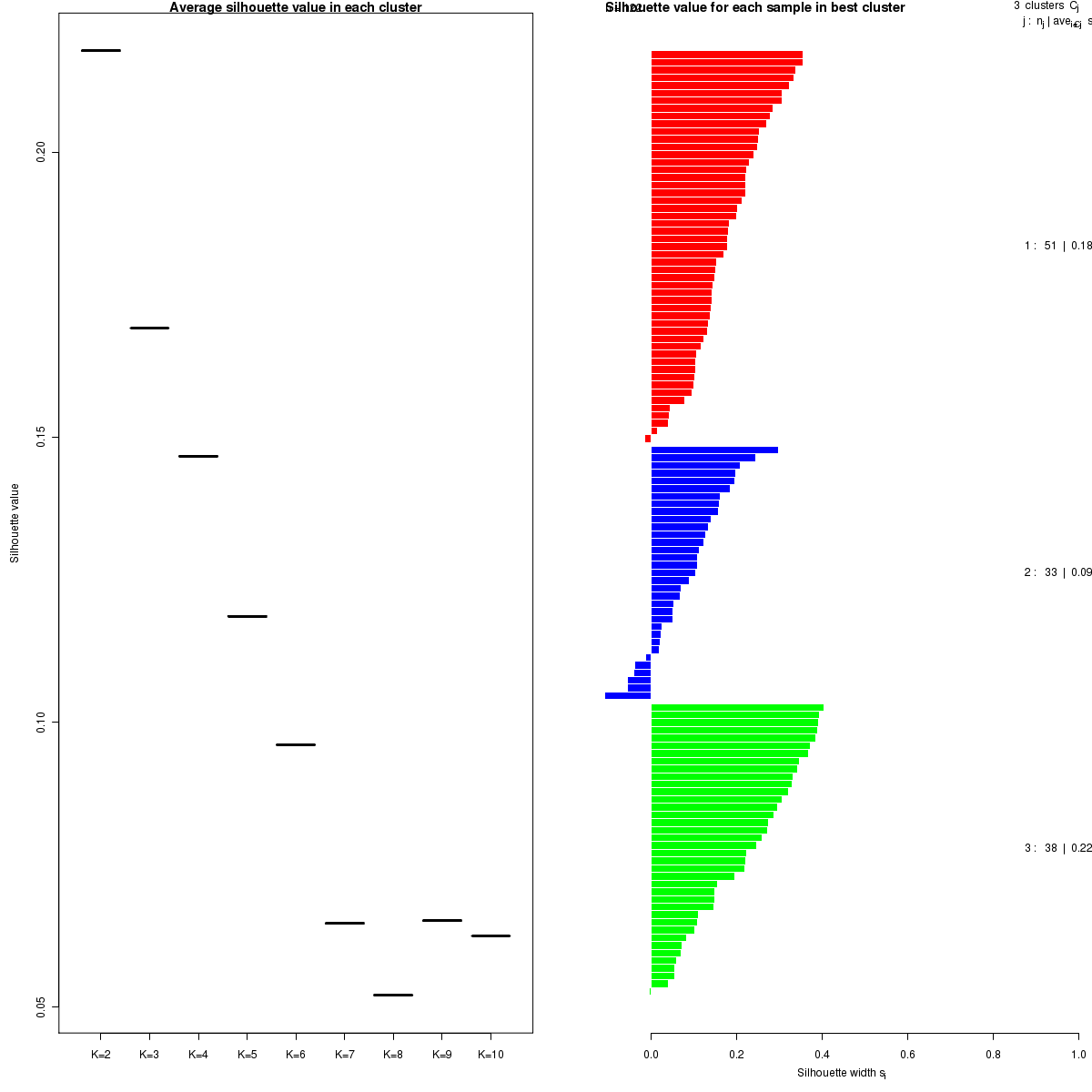
Table 1. Get Full Table List of samples with 3 subtypes and silhouette width.
| SampleName | cluster | silhouetteValue |
|---|---|---|
| TCGA-BL-A0C8-01 | 1 | 0.18 |
| TCGA-BL-A13J-01 | 1 | -0.016 |
| TCGA-BT-A20P-01 | 1 | 0.25 |
| TCGA-BT-A20Q-01 | 1 | 0.014 |
| TCGA-BT-A20T-01 | 1 | 0.1 |
| TCGA-BT-A20W-01 | 1 | 0.21 |
| TCGA-BT-A2LB-01 | 1 | 0.043 |
| TCGA-BT-A3PH-01 | 1 | 0.22 |
| TCGA-C4-A0F6-01 | 1 | 0.15 |
| TCGA-CF-A1HR-01 | 1 | 0.24 |
Table 2. Get Full Table List of samples belonging to each cluster in different k clusters.
| SampleName | K=2 | K=3 | K=4 | K=5 | K=6 | K=7 | K=8 | NA | NA |
|---|---|---|---|---|---|---|---|---|---|
| TCGA-BL-A0C8-01 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| TCGA-BL-A13J-01 | 1 | 1 | 1 | 3 | 3 | 3 | 3 | 1 | 1 |
| TCGA-BT-A20P-01 | 1 | 1 | 1 | 4 | 4 | 5 | 4 | 3 | 6 |
| TCGA-BT-A20Q-01 | 1 | 1 | 1 | 5 | 5 | 6 | 6 | 6 | 6 |
| TCGA-BT-A20R-01 | 1 | 2 | 4 | 5 | 1 | 1 | 1 | 7 | 7 |
| TCGA-BT-A20T-01 | 1 | 1 | 1 | 4 | 3 | 7 | 3 | 1 | 6 |
| TCGA-BT-A20W-01 | 1 | 1 | 1 | 1 | 3 | 7 | 3 | 1 | 1 |
| TCGA-BT-A2LB-01 | 1 | 1 | 1 | 1 | 3 | 7 | 3 | 1 | 1 |
| TCGA-BT-A3PH-01 | 1 | 1 | 1 | 1 | 3 | 7 | 3 | 9 | 9 |
| TCGA-C4-A0F6-01 | 1 | 1 | 1 | 1 | 3 | 7 | 3 | 1 | 1 |
Samples most representative of the clusters, hereby called core samples were identified based on positive silhouette width, indicating higher similarity to their own class than to any other class member. Core samples were used to select differentially expressed marker genes for each subtype by comparing the subclass versus the other subclasses, using Student's t-test.
Table 3. Get Full Table List of marker genes with p <= 0.05 (The positive value of column difference means gene is upregulated in this subtype and vice versa).
| Composite.Element.REF | p | difference | q | subclass |
|---|---|---|---|---|
| HMGCS2|3158 | 6.8e-15 | 7.2 | 2.7e-12 | 1 |
| SPINK1|6690 | 2.8e-12 | 6.6 | 3.1e-10 | 1 |
| DHRS2|10202 | 6.4e-24 | 6.4 | 1.2e-19 | 1 |
| BTBD16|118663 | 6.3e-18 | 6.3 | 1e-14 | 1 |
| UPK1A|11045 | 1.5e-14 | 6 | 4.5e-12 | 1 |
| SNX31|169166 | 3.7e-18 | 5.9 | 7.5e-15 | 1 |
| PSCA|8000 | 3e-13 | 5.7 | 4.9e-11 | 1 |
| SLC14A1|6563 | 3.1e-21 | 5.7 | 1.4e-17 | 1 |
| LOC100188947|100188947 | 2.3e-21 | 5.6 | 1.4e-17 | 1 |
| UPK2|7379 | 1.6e-13 | 5.5 | 3e-11 | 1 |
mRNAseq of normalized RSEM/RPKM value with log2 transformed was as the input RNAseq data for the clustering.
RSEM##REF##5 is used to estimate gene and transcript abundances and these values are normalized to a fixed upper quaritile value of 1000 for gene and 300 for transcript level estimates.
RPKM for a given GeneX is calculated by: (raw read counts * 10^9) / (total reads * length of GeneX). Total reads is the lane yield after removing poor quality reads and the length of GeneX is defined as the median length of all transcripts associated with GeneX.
Non-negative matrix factorization (NMF) is an unsupervised learning algorithm that has been shown to identify molecular patterns when applied to gene expression data ##REF##1,##REF##3. Rather than separating gene clusters based on distance computation, NMF detects contextdependent patterns of gene expression in complex biological systems.
We use the cophenetic correlation coefficient ##REF##1 to determine the cluster that yields the most robust clustering. The cophenetic correlation coefficient is computed based on the consensus matrix of the CNMF clustering, and measures how reliably the same samples are assigned to the same cluster across many iterations of the clustering lgorithm with random initializations. The cophenetic correlation coefficient lies between 0 and 1, with higher values indicating more stable cluster assignments. We select the number of clusters k based on the largest observed correlation coefficient for all tested values of k.
Silhouette width is defined as the ratio of average distance of each sample to samples in the same cluster to the smallest distance to samples not in the same cluster. If silhouette width is close to 1, it means that sample is well clustered. If silhouette width is close to -1, it means that sample is misclassified ##REF##2##REF##4.
This is an experimental feature. The full results of the analysis summarized in this report can be downloaded from the TCGA Data Coordination Center.