This report serves to describe the mutational landscape and properties of a given individual set, as well as rank genes and genesets according to mutational significance. MutSig v2.0 was used to generate the results found in this report.
-
Working with individual set: LIHC-TP
-
Number of patients in set: 197
The input for this pipeline is a set of individuals with the following files associated for each:
-
An annotated .maf file describing the mutations called for the respective individual, and their properties.
-
A .wig file that contains information about the coverage of the sample.
-
MAF used for this analysis:LIHC-TP.final_analysis_set.maf
-
Blacklist used for this analysis: pancan_mutation_blacklist.v14.hg19.txt
-
Significantly mutated genes (q ≤ 0.1): 23
-
Mutations seen in COSMIC: 178
-
Significantly mutated genes in COSMIC territory: 10
-
Significantly mutated genesets: 34
-
Significantly mutated genesets: (excluding sig. mutated genes):0
-
Read 198 MAFs of type "maf1"
-
Total number of mutations in input MAFs: 27832
-
After removing 53 blacklisted mutations: 27779
-
After removing 638 noncoding mutations: 27141
-
After collapsing adjacent/redundant mutations: 27140
-
Number of mutations before filtering: 27140
-
After removing 236 mutations outside patient set: 26904
-
After removing 1399 mutations outside gene set: 25505
-
After removing 33 mutations outside category set: 25472
Table 1. Get Full Table Table representing breakdown of mutations by type.
| type | count |
|---|---|
| De_novo_Start_InFrame | 2 |
| De_novo_Start_OutOfFrame | 5 |
| Frame_Shift_Del | 525 |
| Frame_Shift_Ins | 263 |
| In_Frame_Del | 123 |
| In_Frame_Ins | 22 |
| Missense_Mutation | 16268 |
| Nonsense_Mutation | 849 |
| Nonstop_Mutation | 25 |
| Silent | 6163 |
| Splice_Site | 1188 |
| Start_Codon_Del | 2 |
| Start_Codon_Ins | 1 |
| Start_Codon_SNP | 34 |
| Stop_Codon_Del | 2 |
| Total | 25472 |
Table 2. Get Full Table A breakdown of mutation rates per category discovered for this individual set.
| category | n | N | rate | rate_per_mb | relative_rate | exp_ns_s_ratio |
|---|---|---|---|---|---|---|
| transit | 8161 | 6157259362 | 1.3e-06 | 1.3 | 0.42 | 2 |
| C->A | 3605 | 3141918468 | 1.1e-06 | 1.1 | 0.37 | 4.4 |
| flip | 3570 | 6157259362 | 5.8e-07 | 0.58 | 0.18 | 5.3 |
| A->C | 966 | 3015340894 | 3.2e-07 | 0.32 | 0.1 | 5.2 |
| indel+null | 2975 | 6157259362 | 4.8e-07 | 0.48 | 0.15 | NaN |
| double_null | 32 | 6157259362 | 5.2e-09 | 0.0052 | 0.0017 | NaN |
| Total | 19309 | 6157259362 | 3.1e-06 | 3.1 | 1 | 3.5 |
The x axis represents the samples. The y axis represents the exons, one row per exon, and they are sorted by average coverage across samples. For exons with exactly the same average coverage, they are sorted next by the %GC of the exon. (The secondary sort is especially useful for the zero-coverage exons at the bottom). If the figure is unpopulated, then full coverage is assumed (e.g. MutSig CV doesn't use WIGs and assumes full coverage).
Figure 1.
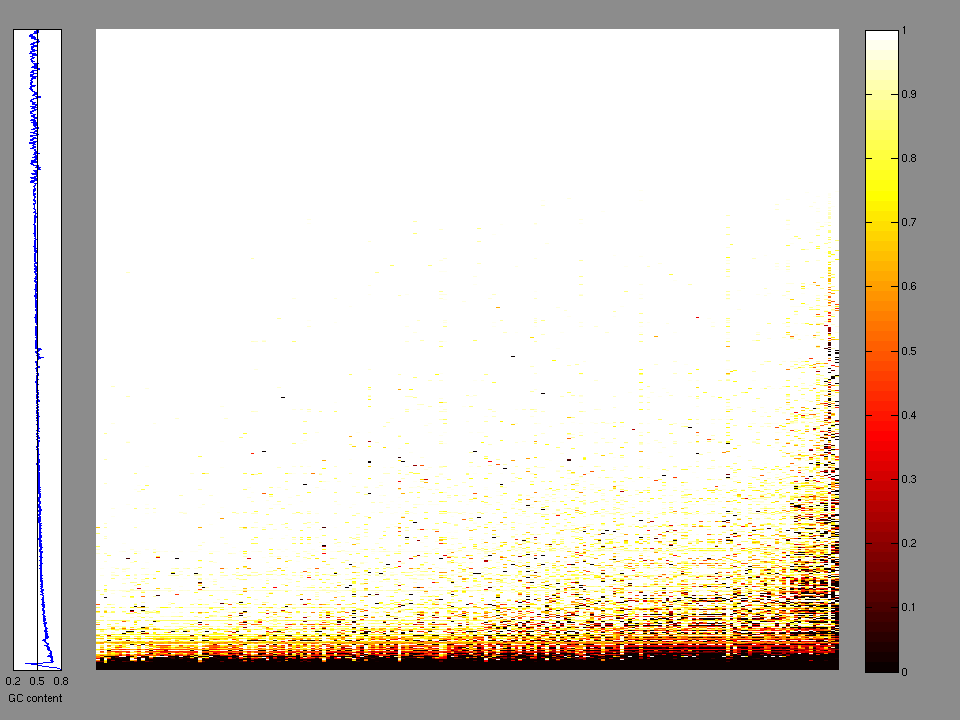
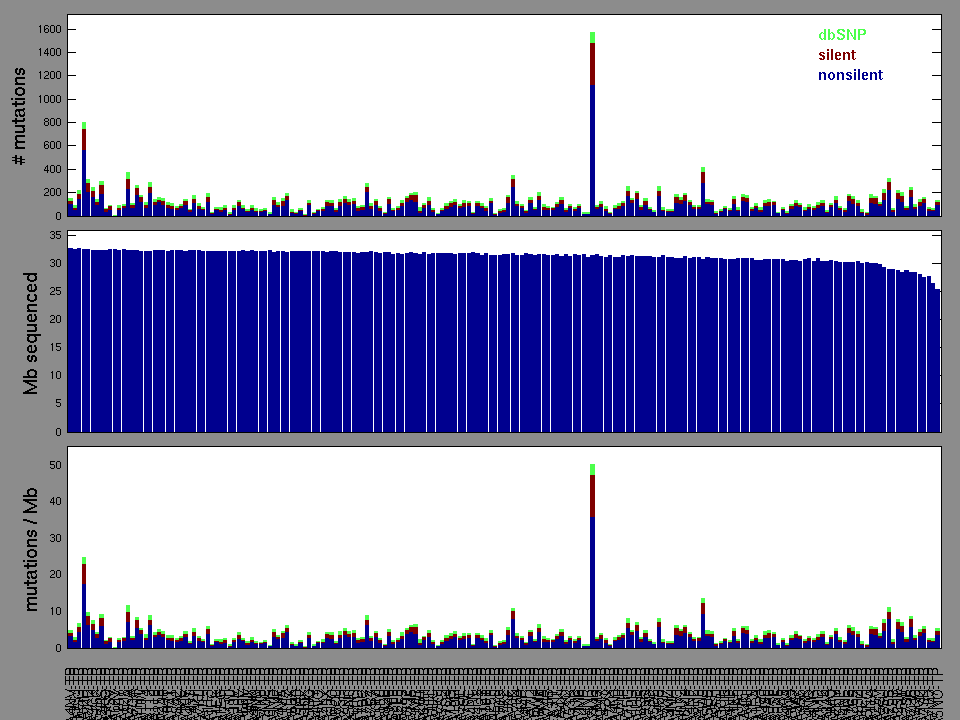
Figure 2. Patients counts and rates file used to generate this plot: LIHC-TP.patients.counts_and_rates.txt
The mutation spectrum is depicted in the lego plots below in which the 96 possible mutation types are subdivided into six large blocks, color-coded to reflect the base substitution type. Each large block is further subdivided into the 16 possible pairs of 5' and 3' neighbors, as listed in the 4x4 trinucleotide context legend. The height of each block corresponds to the mutation frequency for that kind of mutation (counts of mutations normalized by the base coverage in a given bin). The shape of the spectrum is a signature for dominant mutational mechanisms in different tumor types.
Figure 3. Get High-res Image SNV Mutation rate lego plot for entire set. Each bin is normalized by base coverage for that bin. Colors represent the six SNV types on the upper right. The three-base context for each mutation is labeled in the 4x4 legend on the lower right. The fractional breakdown of SNV counts is shown in the pie chart on the upper left. If this figure is blank, not enough information was provided in the MAF to generate it.

Figure 4. Get High-res Image SNV Mutation rate lego plots for 4 slices of mutation allele fraction (0<=AF<0.1, 0.1<=AF<0.25, 0.25<=AF<0.5, & 0.5<=AF) . The color code and three-base context legends are the same as the previous figure. If this figure is blank, not enough information was provided in the MAF to generate it.

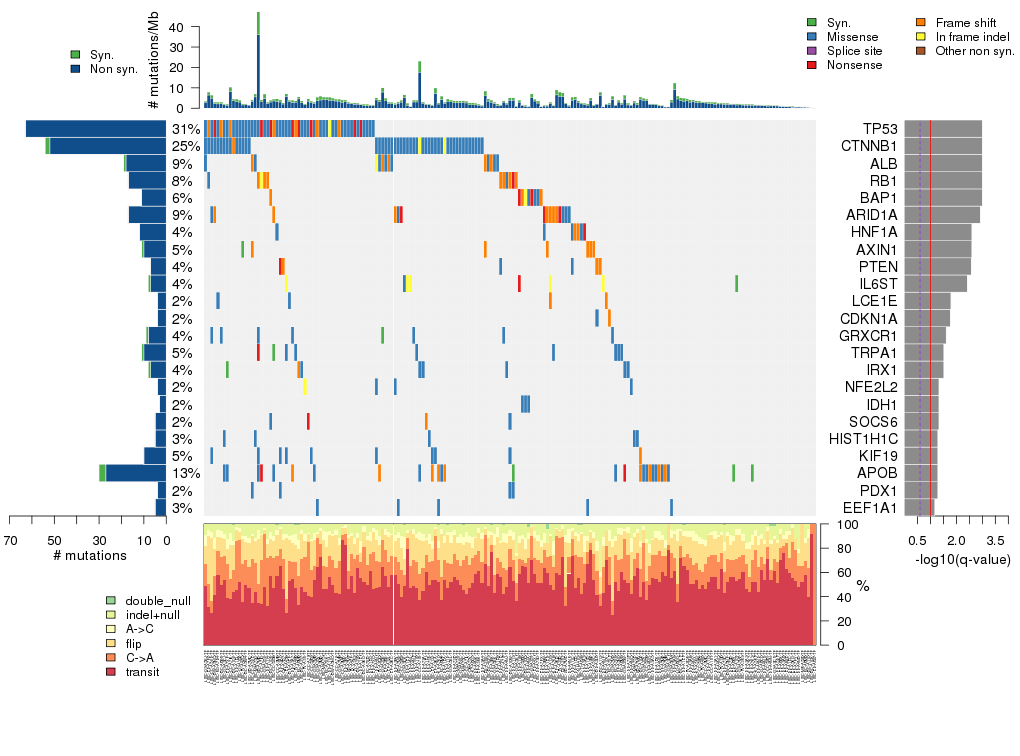
Figure 5. Get High-res Image The matrix in the center of the figure represents individual mutations in patient samples, color-coded by type of mutation, for the significantly mutated genes. The rate of synonymous and non-synonymous mutations is displayed at the top of the matrix. The barplot on the left of the matrix shows the number of mutations in each gene. The percentages represent the fraction of tumors with at least one mutation in the specified gene. The barplot to the right of the matrix displays the q-values for the most significantly mutated genes. The purple boxplots below the matrix (only displayed if required columns are present in the provided MAF) represent the distributions of allelic fractions observed in each sample. The plot at the bottom represents the base substitution distribution of individual samples, using the same categories that were used to calculate significance.
Column Descriptions:
-
N = number of sequenced bases in this gene across the individual set
-
n = number of (nonsilent) mutations in this gene across the individual set
-
npat = number of patients (individuals) with at least one nonsilent mutation
-
nsite = number of unique sites having a non-silent mutation
-
nsil = number of silent mutations in this gene across the individual set
-
n1 = number of nonsilent mutations of type: transit
-
n2 = number of nonsilent mutations of type: C->A
-
n3 = number of nonsilent mutations of type: flip
-
n4 = number of nonsilent mutations of type: A->C
-
n5 = number of nonsilent mutations of type: indel+null
-
n6 = number of nonsilent mutations of type: double_null
-
p_classic = p-value for the observed amount of nonsilent mutations being elevated in this gene
-
p_ns_s = p-value for the observed nonsilent/silent ratio being elevated in this gene
-
p_cons = p-value for enrichment of mutations at evolutionarily most-conserved sites in gene
-
p_joint = p-value for clustering + conservation
-
p = p-value (overall)
-
q = q-value, False Discovery Rate (Benjamini-Hochberg procedure)
Table 3. Get Full Table A Ranked List of Significantly Mutated Genes. Number of significant genes found: 23. Number of genes displayed: 35. Click on a gene name to display its stick figure depicting the distribution of mutations and mutation types across the chosen gene (this feature may not be available for all significant genes).
| rank | gene | description | N | n | npat | nsite | nsil | n1 | n2 | n3 | n4 | n5 | n6 | p_classic | p_ns_s | p_clust | p_cons | p_joint | p | q |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | TP53 | tumor protein p53 | 249557 | 63 | 62 | 50 | 0 | 19 | 15 | 3 | 2 | 24 | 0 | 2e-15 | 8.5e-08 | 0 | 0.0027 | 0 | <1.00e-15 | <9.32e-12 |
| 2 | CTNNB1 | catenin (cadherin-associated protein), beta 1, 88kDa | 471692 | 52 | 50 | 25 | 2 | 20 | 4 | 18 | 7 | 2 | 1 | 4.8e-15 | 0.000066 | 0 | 0.05 | 0 | <1.00e-15 | <9.32e-12 |
| 3 | ALB | albumin | 369854 | 18 | 18 | 18 | 1 | 5 | 1 | 1 | 1 | 7 | 3 | 1e-14 | 0.18 | 0.37 | 0.7 | 0.51 | 1.81e-13 | 1.13e-09 |
| 4 | RB1 | retinoblastoma 1 (including osteosarcoma) | 518944 | 17 | 15 | 17 | 0 | 0 | 0 | 2 | 1 | 13 | 1 | 9.7e-13 | 0.085 | 0.0059 | 0.78 | 0.014 | 4.65e-13 | 2.17e-09 |
| 5 | BAP1 | BRCA1 associated protein-1 (ubiquitin carboxy-terminal hydrolase) | 435698 | 11 | 11 | 11 | 0 | 0 | 0 | 1 | 2 | 8 | 0 | 1.5e-09 | 0.22 | 0.025 | 0.11 | 0.018 | 6.74e-10 | 2.51e-06 |
| 6 | ARID1A | AT rich interactive domain 1A (SWI-like) | 1137771 | 17 | 17 | 16 | 0 | 1 | 0 | 3 | 1 | 11 | 1 | 2.8e-09 | 0.064 | 0.57 | 0.53 | 1 | 5.78e-08 | 0.000180 |
| 7 | HNF1A | HNF1 homeobox A | 356985 | 12 | 8 | 12 | 0 | 4 | 1 | 3 | 0 | 4 | 0 | 1.7e-06 | 0.04 | 0.0095 | 0.83 | 0.019 | 5.86e-07 | 0.00156 |
| 8 | AXIN1 | axin 1 | 434917 | 10 | 9 | 10 | 1 | 0 | 0 | 1 | 0 | 9 | 0 | 2.2e-07 | 0.62 | 0.19 | 0.2 | 0.17 | 6.74e-07 | 0.00157 |
| 9 | PTEN | phosphatase and tensin homolog (mutated in multiple advanced cancers 1) | 237849 | 7 | 7 | 7 | 0 | 1 | 0 | 1 | 0 | 5 | 0 | 2e-06 | 0.47 | 0.01 | 0.8 | 0.022 | 8.03e-07 | 0.00166 |
| 10 | IL6ST | interleukin 6 signal transducer (gp130, oncostatin M receptor) | 553112 | 7 | 7 | 7 | 1 | 0 | 1 | 0 | 0 | 6 | 0 | 0.0001 | 0.84 | 0.00035 | 0.6 | 0.00087 | 1.50e-06 | 0.00280 |
| 11 | LCE1E | late cornified envelope 1E | 70748 | 4 | 4 | 4 | 0 | 1 | 1 | 0 | 0 | 2 | 0 | 9.8e-06 | 0.57 | 0.035 | 0.93 | 0.06 | 9.03e-06 | 0.0153 |
| 12 | CDKN1A | cyclin-dependent kinase inhibitor 1A (p21, Cip1) | 87268 | 4 | 4 | 4 | 0 | 0 | 1 | 0 | 1 | 2 | 0 | 4.1e-06 | 0.49 | 0.94 | 0.035 | 0.16 | 1.03e-05 | 0.0160 |
| 13 | GRXCR1 | glutaredoxin, cysteine rich 1 | 174143 | 8 | 7 | 8 | 1 | 2 | 3 | 3 | 0 | 0 | 0 | 1.1e-06 | 0.37 | 0.7 | 0.7 | 1 | 1.64e-05 | 0.0235 |
| 14 | TRPA1 | transient receptor potential cation channel, subfamily A, member 1 | 669092 | 10 | 10 | 10 | 1 | 1 | 4 | 2 | 0 | 3 | 0 | 7.2e-06 | 0.48 | 0.22 | 0.2 | 0.22 | 2.24e-05 | 0.0297 |
| 15 | IRX1 | iroquois homeobox 1 | 195940 | 7 | 7 | 7 | 1 | 2 | 3 | 1 | 0 | 1 | 0 | 3e-06 | 0.43 | 0.86 | 0.26 | 0.56 | 2.39e-05 | 0.0297 |
| 16 | NFE2L2 | nuclear factor (erythroid-derived 2)-like 2 | 358226 | 4 | 4 | 4 | 0 | 0 | 0 | 1 | 2 | 1 | 0 | 0.004 | 0.58 | 0.0035 | 0.013 | 0.00076 | 4.20e-05 | 0.0459 |
| 17 | IDH1 | isocitrate dehydrogenase 1 (NADP+), soluble | 250729 | 3 | 3 | 1 | 0 | 2 | 0 | 1 | 0 | 0 | 0 | 0.011 | 0.4 | 9e-05 | 0.69 | 0.0003 | 4.38e-05 | 0.0459 |
| 18 | SOCS6 | suppressor of cytokine signaling 6 | 312825 | 5 | 4 | 5 | 0 | 1 | 0 | 1 | 0 | 3 | 0 | 0.0059 | 0.36 | 0.00031 | 0.46 | 0.00055 | 4.43e-05 | 0.0459 |
| 19 | HIST1H1C | histone cluster 1, H1c | 126299 | 5 | 5 | 5 | 0 | 4 | 0 | 1 | 0 | 0 | 0 | 0.000027 | 0.15 | 0.068 | 0.9 | 0.15 | 5.51e-05 | 0.0518 |
| 20 | KIF19 | kinesin family member 19 | 499210 | 10 | 10 | 10 | 0 | 6 | 2 | 0 | 1 | 1 | 0 | 5.8e-06 | 0.042 | 0.51 | 0.46 | 0.74 | 5.65e-05 | 0.0518 |
| 21 | APOB | apolipoprotein B (including Ag(x) antigen) | 2679262 | 27 | 25 | 27 | 3 | 9 | 3 | 4 | 0 | 11 | 0 | 7e-05 | 0.19 | 0.24 | 0.047 | 0.062 | 5.83e-05 | 0.0518 |
| 22 | PDX1 | pancreatic and duodenal homeobox 1 | 80852 | 4 | 4 | 4 | 0 | 2 | 2 | 0 | 0 | 0 | 0 | 0.000081 | 0.27 | 0.59 | 0.013 | 0.057 | 6.11e-05 | 0.0518 |
| 23 | EEF1A1 | eukaryotic translation elongation factor 1 alpha 1 | 220690 | 5 | 5 | 2 | 0 | 5 | 0 | 0 | 0 | 0 | 0 | 0.0005 | 0.14 | 0.021 | 0.028 | 0.013 | 8.30e-05 | 0.0673 |
| 24 | DCDC1 | doublecortin domain containing 1 | 213661 | 6 | 5 | 6 | 0 | 3 | 1 | 0 | 0 | 2 | 0 | 0.00015 | 0.17 | NaN | NaN | NaN | 0.000147 | 0.114 |
| 25 | SPRR4 | small proline-rich protein 4 | 48053 | 3 | 3 | 3 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 0.00025 | 0.49 | 0.29 | 0.027 | 0.051 | 0.000158 | 0.118 |
| 26 | SGCZ | sarcoglycan zeta | 184196 | 5 | 5 | 5 | 0 | 1 | 1 | 1 | 0 | 2 | 0 | 0.000022 | 0.21 | 0.5 | 0.99 | 0.63 | 0.000166 | 0.119 |
| 27 | CHL1 | cell adhesion molecule with homology to L1CAM (close homolog of L1) | 731382 | 11 | 11 | 11 | 1 | 5 | 3 | 2 | 1 | 0 | 0 | 0.000016 | 0.21 | 0.97 | 0.59 | 1 | 0.000199 | 0.137 |
| 28 | MAB21L1 | mab-21-like 1 (C. elegans) | 206986 | 6 | 6 | 6 | 0 | 3 | 2 | 1 | 0 | 0 | 0 | 5e-05 | 0.13 | 0.38 | 0.54 | 0.47 | 0.000277 | 0.185 |
| 29 | KEAP1 | kelch-like ECH-associated protein 1 | 355860 | 8 | 7 | 8 | 0 | 4 | 3 | 0 | 0 | 1 | 0 | 0.00011 | 0.084 | 0.2 | 0.2 | 0.23 | 0.000294 | 0.186 |
| 30 | IL12A | interleukin 12A (natural killer cell stimulatory factor 1, cytotoxic lymphocyte maturation factor 1, p35) | 153415 | 5 | 5 | 5 | 0 | 2 | 1 | 2 | 0 | 0 | 0 | 6e-05 | 0.26 | 0.2 | 0.83 | 0.43 | 0.000299 | 0.186 |
| 31 | SCAPER | S phase cyclin A-associated protein in the ER | 836833 | 8 | 8 | 8 | 0 | 5 | 0 | 2 | 0 | 1 | 0 | 0.0047 | 0.086 | 0.091 | 0.0042 | 0.0083 | 0.000434 | 0.261 |
| 32 | ZNF676 | zinc finger protein 676 | 324631 | 7 | 6 | 7 | 0 | 0 | 2 | 3 | 0 | 2 | 0 | 0.00016 | 0.52 | 0.12 | 0.99 | 0.25 | 0.000448 | 0.261 |
| 33 | LDOC1L | leucine zipper, down-regulated in cancer 1-like | 135746 | 5 | 5 | 5 | 0 | 3 | 1 | 0 | 0 | 1 | 0 | 0.000046 | 0.16 | 0.97 | 0.66 | 1 | 0.000503 | 0.284 |
| 34 | CADPS | Ca2+-dependent secretion activator | 791011 | 9 | 9 | 9 | 1 | 3 | 1 | 3 | 0 | 2 | 0 | 0.00045 | 0.31 | 0.13 | 0.11 | 0.11 | 0.000522 | 0.285 |
| 35 | TUSC3 | tumor suppressor candidate 3 | 205941 | 5 | 4 | 5 | 0 | 1 | 2 | 1 | 1 | 0 | 0 | 0.0028 | 0.32 | 0.0087 | 0.96 | 0.018 | 0.000535 | 0.285 |
Figure S1. This figure depicts the distribution of mutations and mutation types across the TP53 significant gene.

Figure S2. This figure depicts the distribution of mutations and mutation types across the CTNNB1 significant gene.
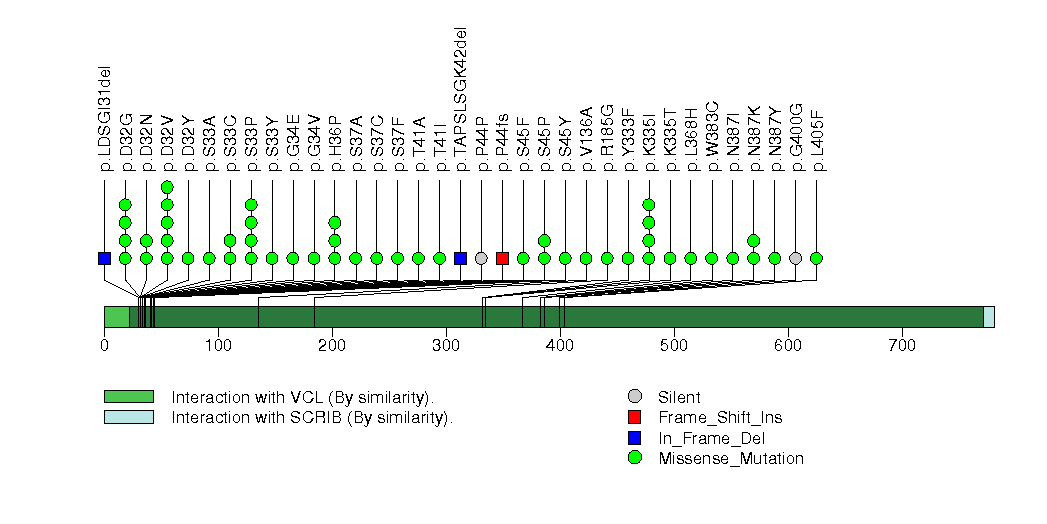
Figure S3. This figure depicts the distribution of mutations and mutation types across the ALB significant gene.
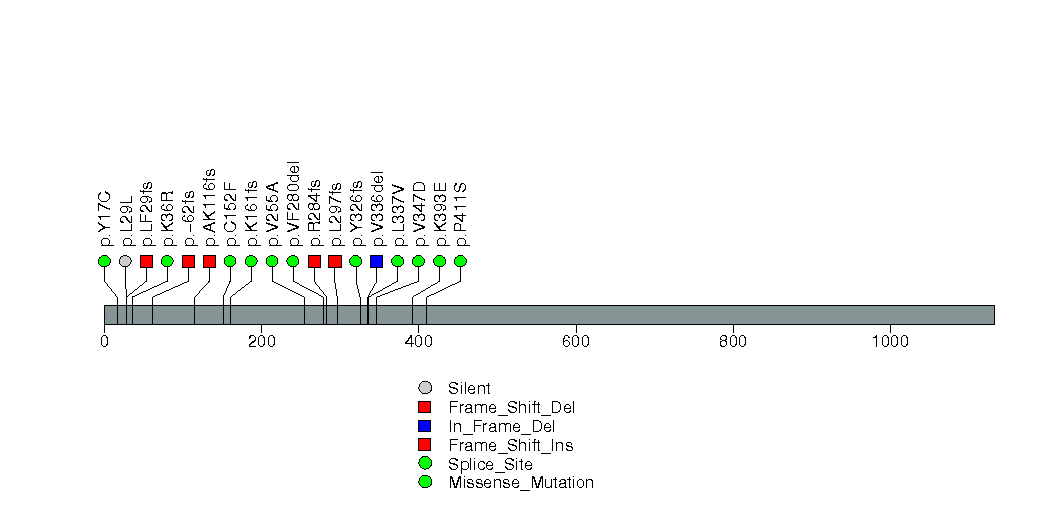
Figure S4. This figure depicts the distribution of mutations and mutation types across the RB1 significant gene.
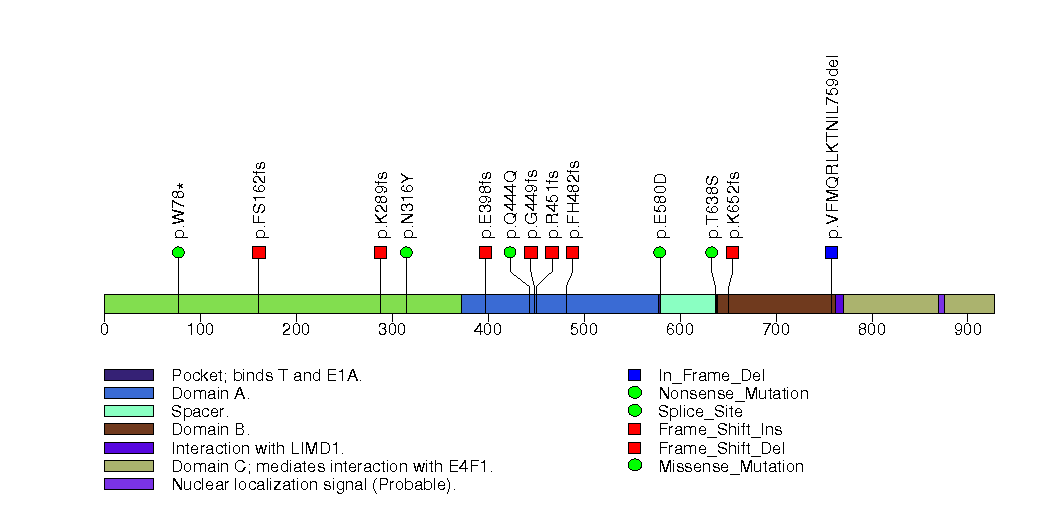
Figure S5. This figure depicts the distribution of mutations and mutation types across the BAP1 significant gene.
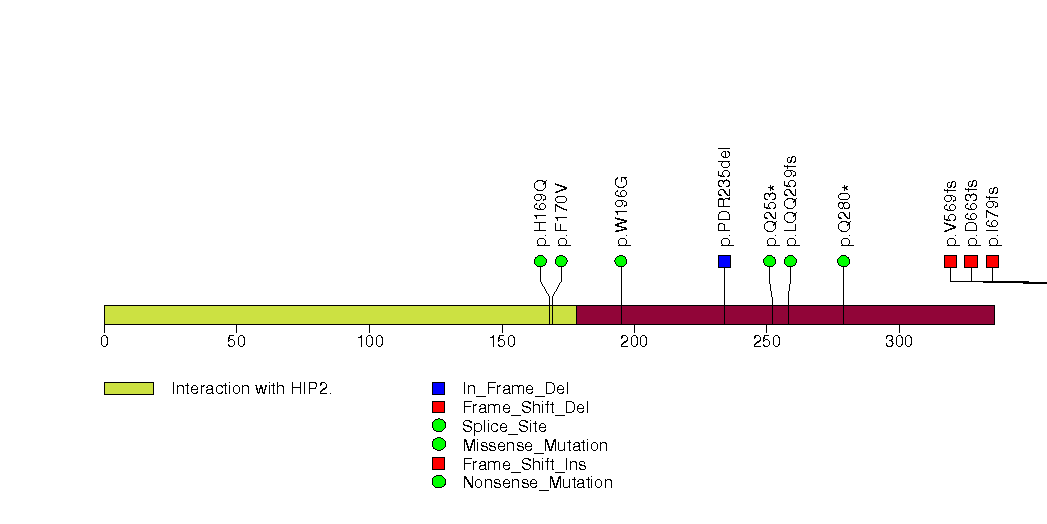
Figure S6. This figure depicts the distribution of mutations and mutation types across the ARID1A significant gene.
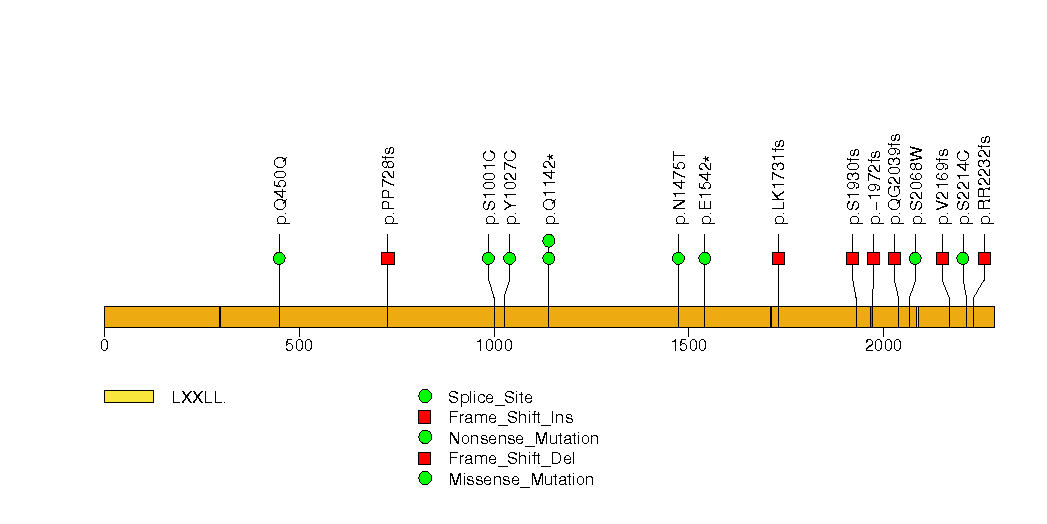
Figure S7. This figure depicts the distribution of mutations and mutation types across the HNF1A significant gene.
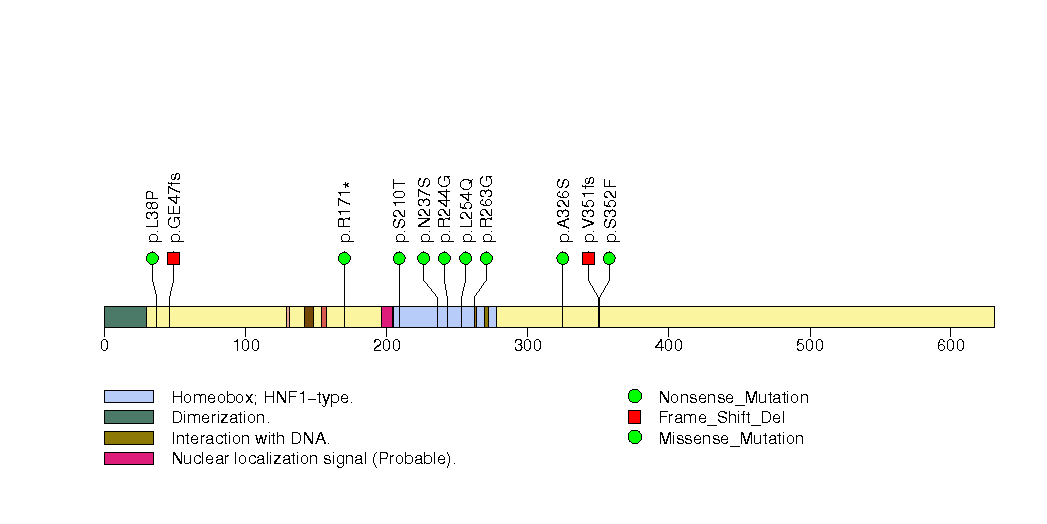
Figure S8. This figure depicts the distribution of mutations and mutation types across the PTEN significant gene.
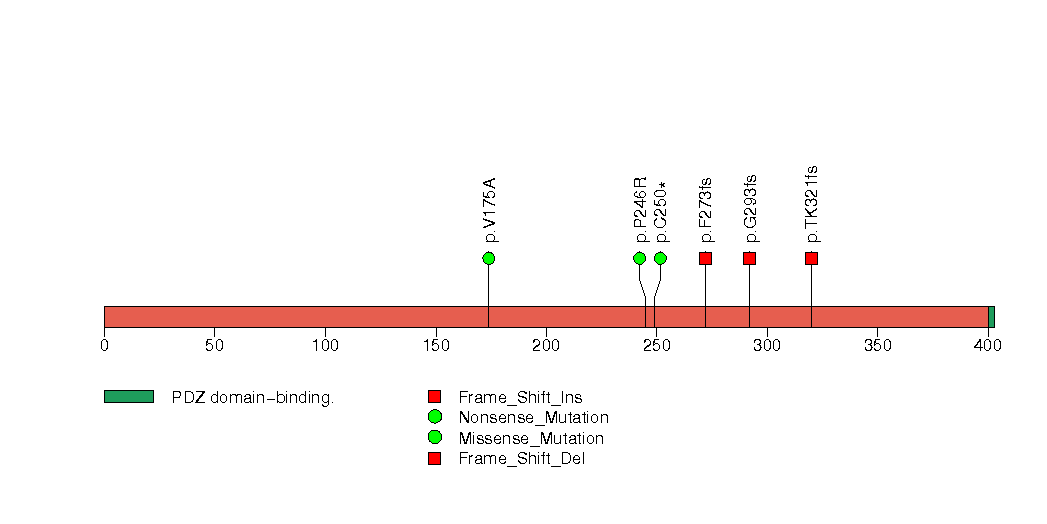
Figure S9. This figure depicts the distribution of mutations and mutation types across the IL6ST significant gene.
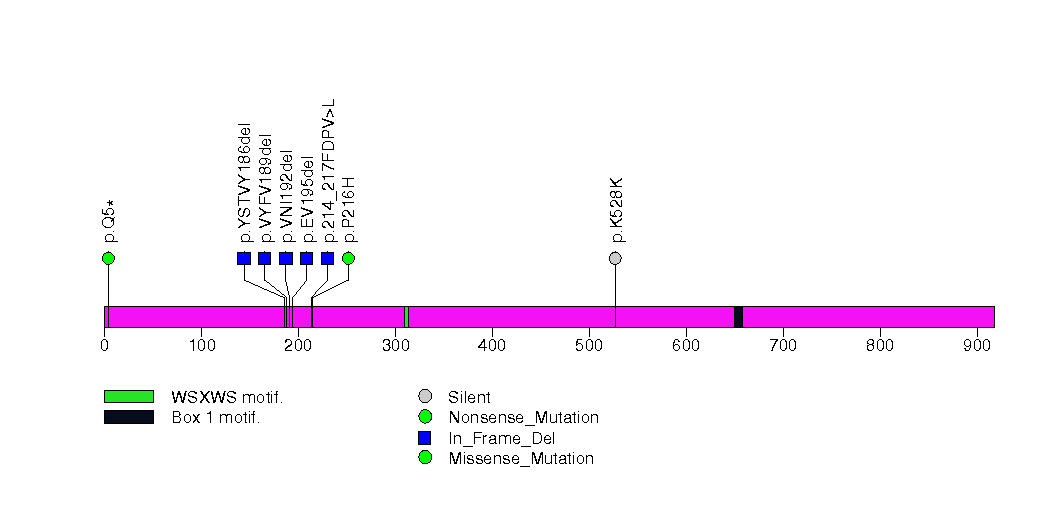
Figure S10. This figure depicts the distribution of mutations and mutation types across the LCE1E significant gene.
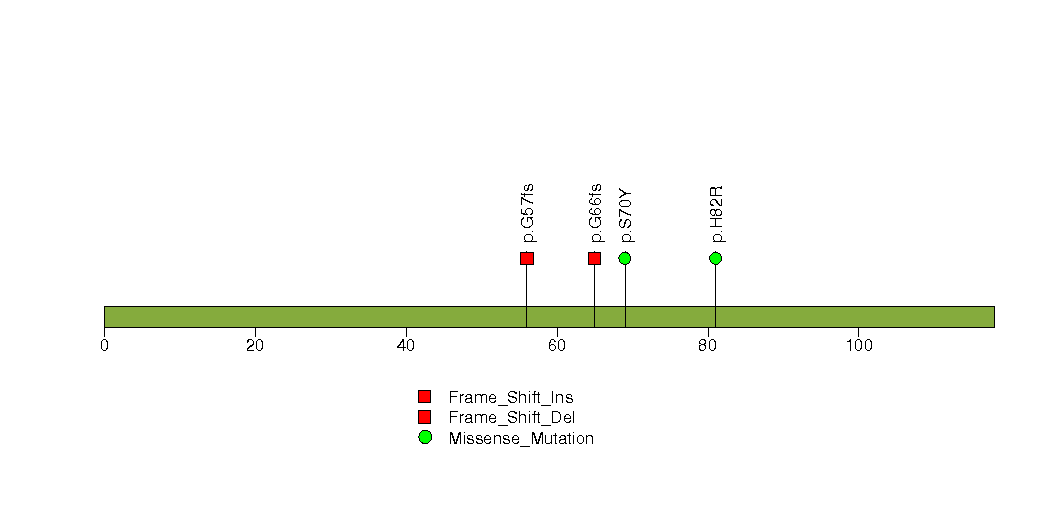
Figure S11. This figure depicts the distribution of mutations and mutation types across the CDKN1A significant gene.
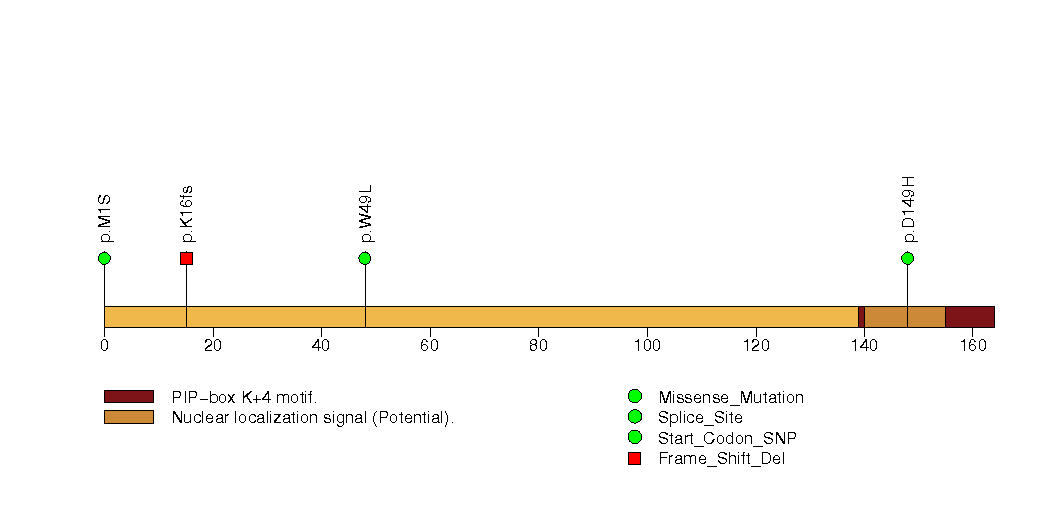
Figure S12. This figure depicts the distribution of mutations and mutation types across the GRXCR1 significant gene.
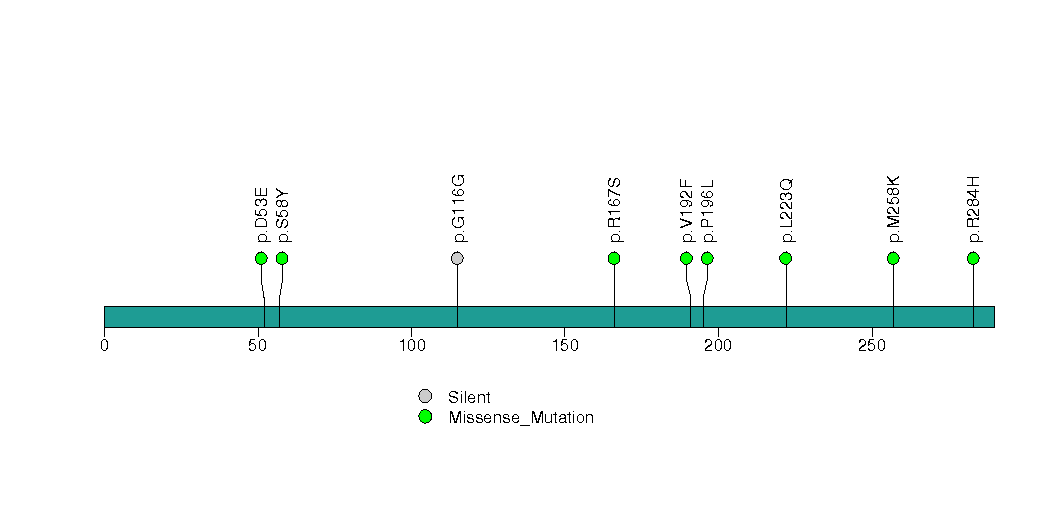
Figure S13. This figure depicts the distribution of mutations and mutation types across the TRPA1 significant gene.
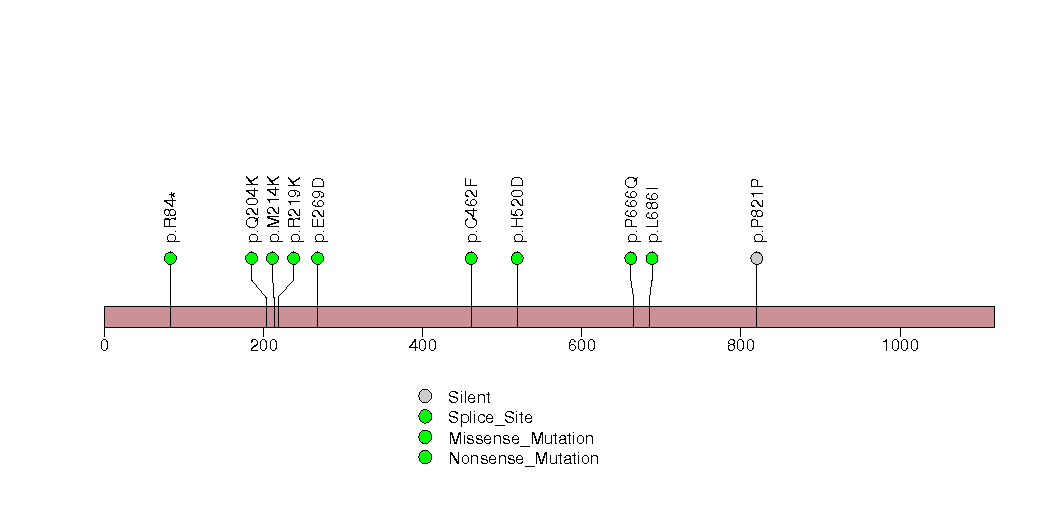
Figure S14. This figure depicts the distribution of mutations and mutation types across the IRX1 significant gene.
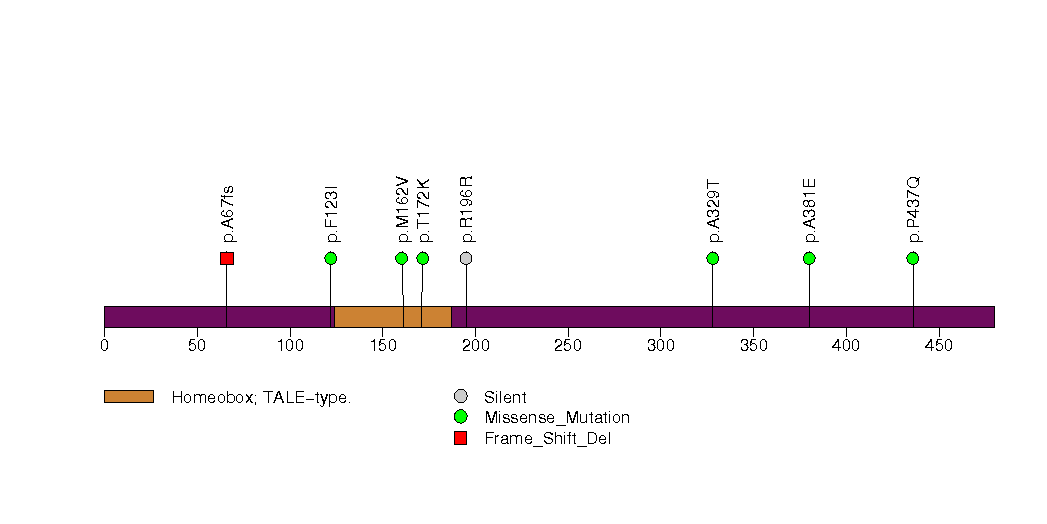
Figure S15. This figure depicts the distribution of mutations and mutation types across the NFE2L2 significant gene.
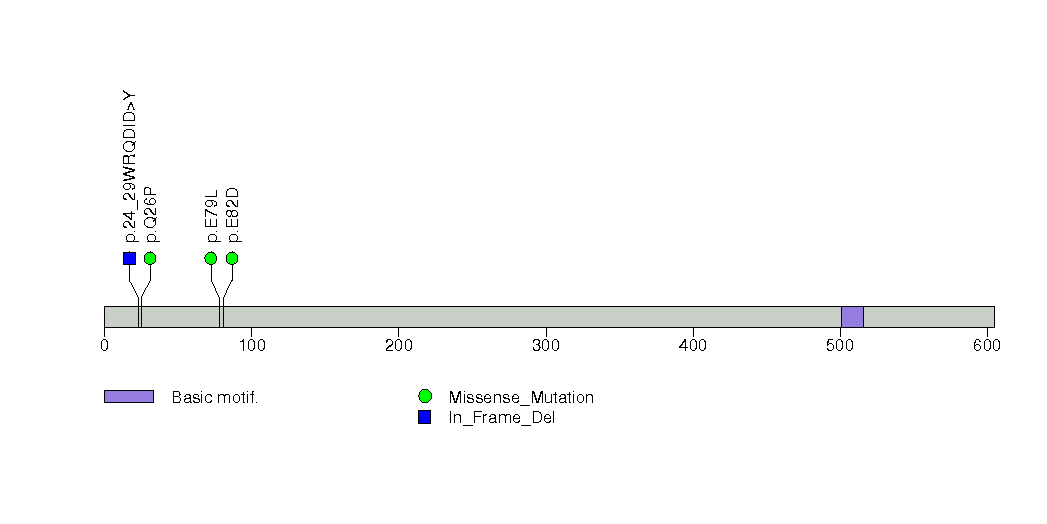
Figure S16. This figure depicts the distribution of mutations and mutation types across the IDH1 significant gene.
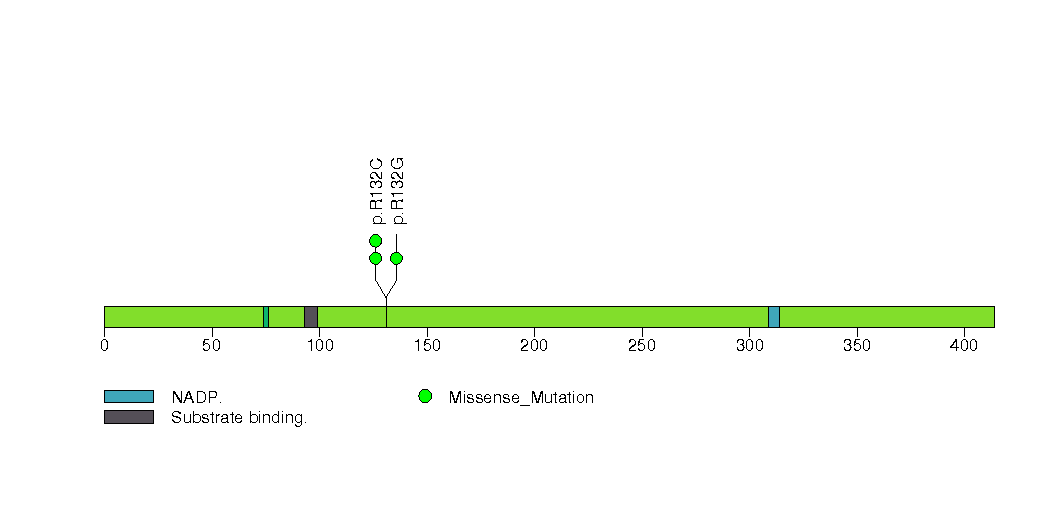
Figure S17. This figure depicts the distribution of mutations and mutation types across the SOCS6 significant gene.
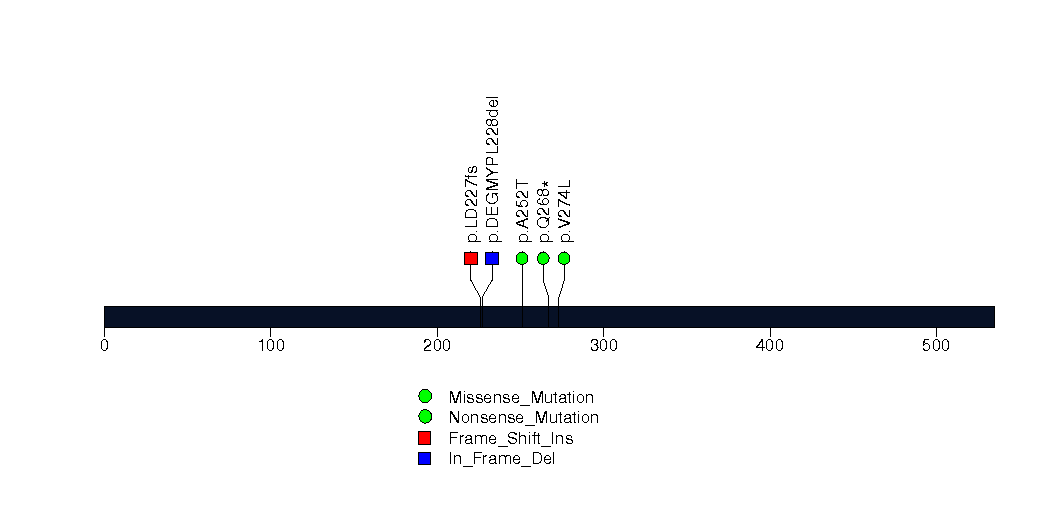
Figure S18. This figure depicts the distribution of mutations and mutation types across the HIST1H1C significant gene.
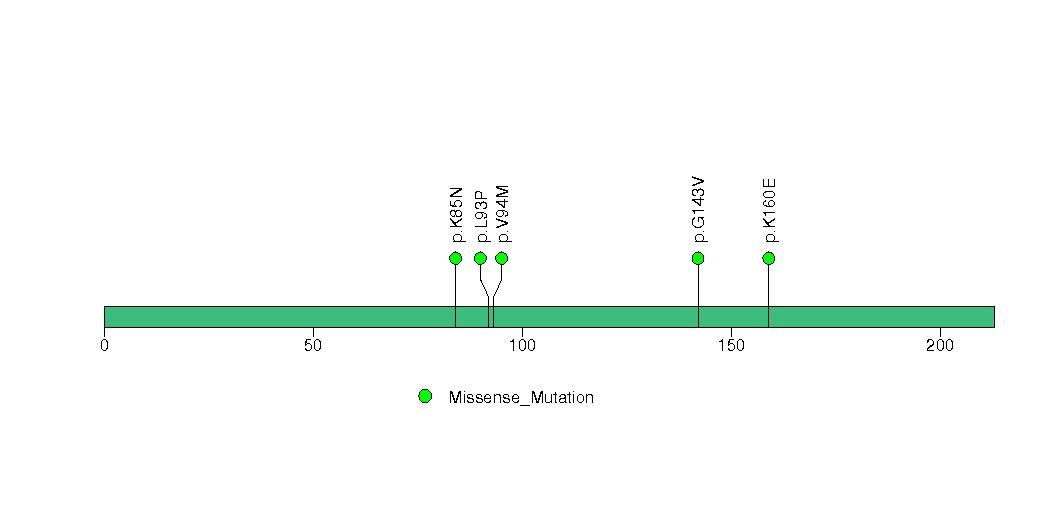
Figure S19. This figure depicts the distribution of mutations and mutation types across the KIF19 significant gene.
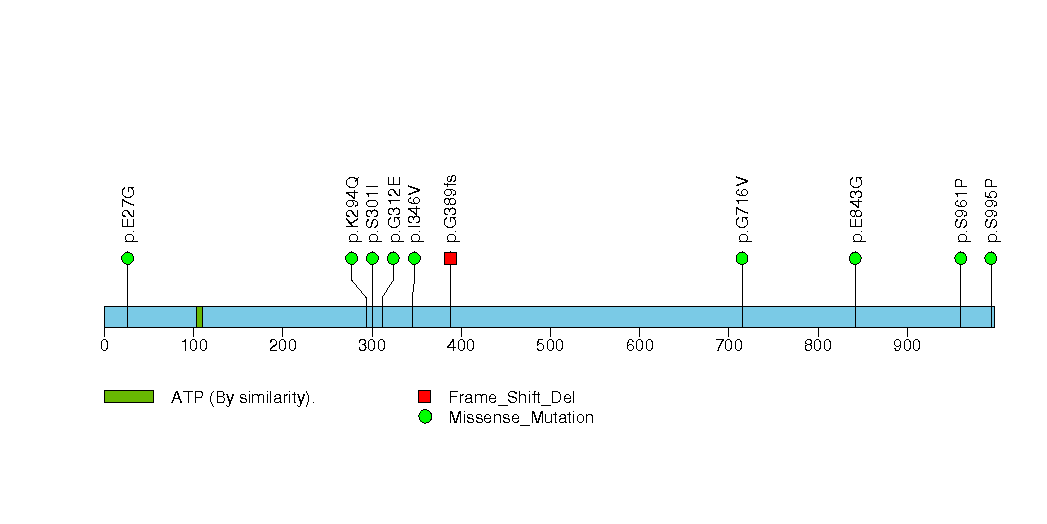
Figure S20. This figure depicts the distribution of mutations and mutation types across the APOB significant gene.
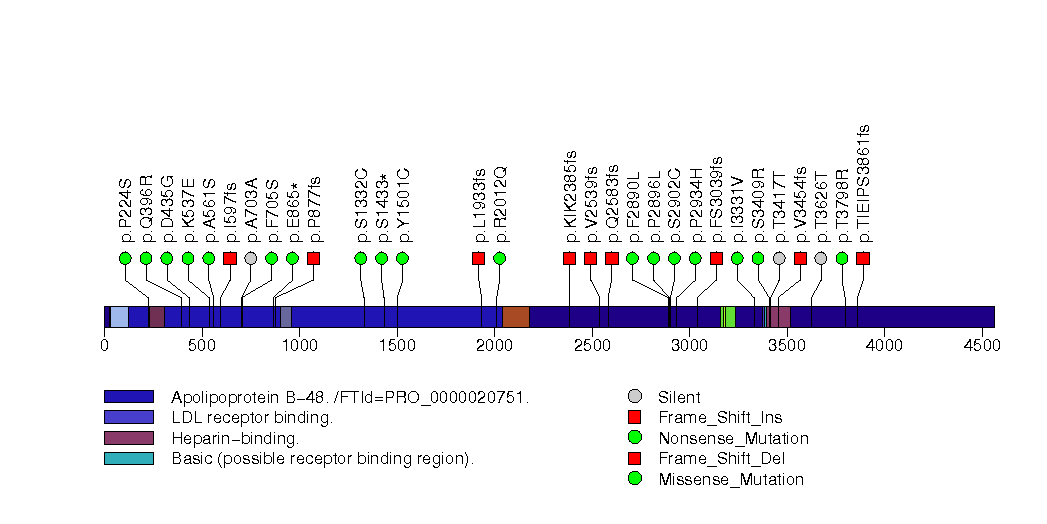
Figure S21. This figure depicts the distribution of mutations and mutation types across the PDX1 significant gene.
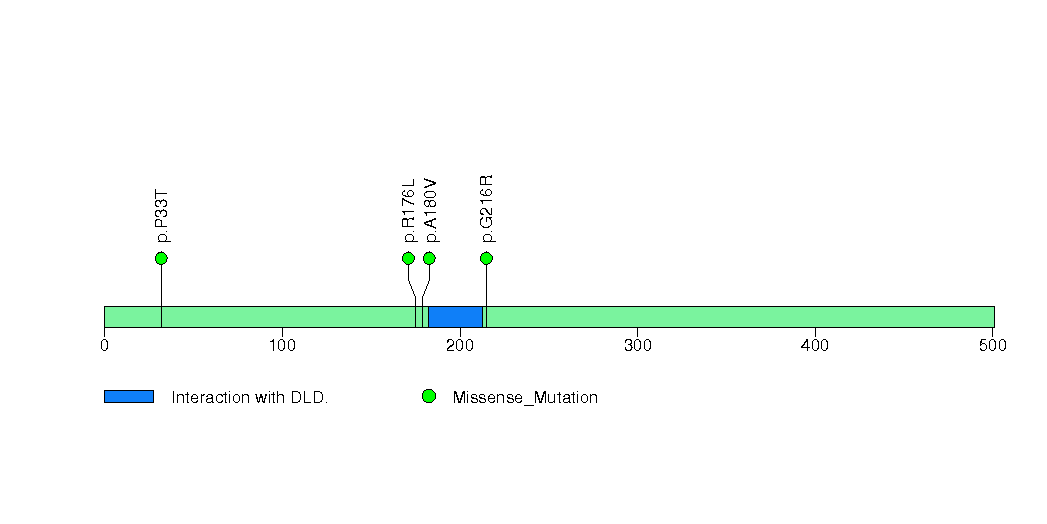
Figure S22. This figure depicts the distribution of mutations and mutation types across the EEF1A1 significant gene.
In this analysis, COSMIC is used as a filter to increase power by restricting the territory of each gene. Cosmic version: v48.
Table 4. Get Full Table Significantly mutated genes (COSMIC territory only). To access the database please go to: COSMIC. Number of significant genes found: 10. Number of genes displayed: 10
| rank | gene | description | n | cos | n_cos | N_cos | cos_ev | p | q |
|---|---|---|---|---|---|---|---|---|---|
| 1 | CTNNB1 | catenin (cadherin-associated protein), beta 1, 88kDa | 52 | 138 | 42 | 27186 | 11963 | 8.1e-13 | 2.8e-09 |
| 2 | RB1 | retinoblastoma 1 (including osteosarcoma) | 17 | 267 | 13 | 52599 | 27 | 1.4e-12 | 2.8e-09 |
| 3 | TP53 | tumor protein p53 | 63 | 356 | 59 | 70132 | 10382 | 1.8e-12 | 2.8e-09 |
| 4 | IDH1 | isocitrate dehydrogenase 1 (NADP+), soluble | 3 | 5 | 3 | 985 | 4476 | 4.9e-09 | 5.5e-06 |
| 5 | GNAS | GNAS complex locus | 7 | 7 | 3 | 1379 | 630 | 1.3e-08 | 0.000012 |
| 6 | PIK3CA | phosphoinositide-3-kinase, catalytic, alpha polypeptide | 7 | 220 | 5 | 43340 | 2949 | 3.5e-07 | 0.00026 |
| 7 | PTEN | phosphatase and tensin homolog (mutated in multiple advanced cancers 1) | 7 | 767 | 7 | 151099 | 82 | 7e-07 | 0.00046 |
| 8 | KRAS | v-Ki-ras2 Kirsten rat sarcoma viral oncogene homolog | 4 | 52 | 3 | 10244 | 16864 | 5.4e-06 | 0.0031 |
| 9 | IDH2 | isocitrate dehydrogenase 2 (NADP+), mitochondrial | 4 | 6 | 2 | 1182 | 166 | 6.8e-06 | 0.0035 |
| 10 | HNF1A | HNF1 homeobox A | 12 | 98 | 3 | 19306 | 8 | 0.000035 | 0.016 |
Note:
n - number of (nonsilent) mutations in this gene across the individual set.
cos = number of unique mutated sites in this gene in COSMIC
n_cos = overlap between n and cos.
N_cos = number of individuals times cos.
cos_ev = total evidence: number of reports in COSMIC for mutations seen in this gene.
p = p-value for seeing the observed amount of overlap in this gene)
q = q-value, False Discovery Rate (Benjamini-Hochberg procedure)
Table 5. Get Full Table A Ranked List of Significantly Mutated Genesets. (Source: MSigDB GSEA Cannonical Pathway Set).Number of significant genesets found: 34. Number of genesets displayed: 10
| rank | geneset | description | genes | N_genes | mut_tally | N | n | npat | nsite | nsil | n1 | n2 | n3 | n4 | n5 | n6 | p_ns_s | p | q |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | ARFPATHWAY | Cyclin-dependent kinase inhibitor 2A is a tumor suppressor that induces G1 arrest and can activate the p53 pathway, leading to G2/M arrest. | ABL1, CDKN2A, E2F1, MDM2, MYC, PIK3CA, PIK3R1, POLR1A, POLR1B, POLR1C, POLR1D, RAC1, RB1, TBX2, TP53, TWIST1 | 16 | CDKN2A(1), E2F1(1), MDM2(1), MYC(1), PIK3CA(7), PIK3R1(2), POLR1A(1), RB1(17), TBX2(3), TP53(63) | 5835542 | 97 | 83 | 83 | 3 | 26 | 18 | 8 | 4 | 40 | 1 | 1.7e-07 | 1.1e-15 | 2.1e-13 |
| 2 | TERTPATHWAY | hTERC, the RNA subunit of telomerase, and hTERT, the catalytic protein subunit, are required for telomerase activity and are overexpressed in many cancers. | HDAC1, MAX, MYC, SP1, SP3, TP53, WT1, ZNF42 | 7 | MYC(1), SP1(2), SP3(3), TP53(63), WT1(3) | 2072209 | 72 | 70 | 59 | 0 | 25 | 15 | 4 | 3 | 25 | 0 | 7e-09 | 1.6e-15 | 2.1e-13 |
| 3 | G1PATHWAY | CDK4/6-cyclin D and CDK2-cyclin E phosphorylate Rb, which allows the transcription of genes needed for the G1/S cell cycle transition. | ABL1, ATM, ATR, CCNA1, CCND1, CCNE1, CDC2, CDC25A, CDK2, CDK4, CDK6, CDKN1A, CDKN1B, CDKN2A, CDKN2B, DHFR, E2F1, GSK3B, HDAC1, MADH3, MADH4, RB1, SKP2, TFDP1, TGFB1, TGFB2, TGFB3, TP53 | 25 | ATM(7), ATR(6), CCNA1(1), CCNE1(1), CDKN1A(4), CDKN2A(1), E2F1(1), GSK3B(2), RB1(17), SKP2(1), TFDP1(1), TGFB2(2), TGFB3(2), TP53(63) | 8784671 | 109 | 86 | 96 | 7 | 26 | 19 | 15 | 6 | 42 | 1 | 0.000012 | 1.7e-15 | 2.1e-13 |
| 4 | PS1PATHWAY | Presenilin is required for gamma-secretase activity to activate Notch signaling; presenilin also inhibits beta-catenin in the Wnt/Frizzled pathway. | ADAM17, APC, AXIN1, BTRC, CTNNB1, DLL1, DVL1, FZD1, GSK3B, NOTCH1, PSEN1, RBPSUH, TCF1, WNT1 | 12 | ADAM17(4), APC(5), AXIN1(10), BTRC(1), CTNNB1(52), DVL1(1), FZD1(1), GSK3B(2), NOTCH1(1), PSEN1(1), WNT1(2) | 6301745 | 80 | 72 | 53 | 4 | 28 | 6 | 21 | 9 | 15 | 1 | 7.8e-06 | 1.7e-15 | 2.1e-13 |
| 5 | TIDPATHWAY | On ligand binding, interferon gamma receptors stimulate JAK2 kinase to phosphorylate STAT transcription factors, which promote expression of interferon responsive genes. | DNAJA3, HSPA1A, IFNG, IFNGR1, IFNGR2, IKBKB, JAK2, LIN7A, NFKB1, NFKBIA, RB1, RELA, TIP-1, TNF, TNFRSF1A, TNFRSF1B, TP53, USH1C, WT1 | 18 | IFNG(2), IFNGR1(2), IKBKB(3), JAK2(4), NFKB1(2), NFKBIA(2), RB1(17), RELA(1), TNF(2), TNFRSF1A(1), TP53(63), USH1C(2), WT1(3) | 5649068 | 104 | 83 | 91 | 3 | 30 | 18 | 11 | 4 | 40 | 1 | 2.2e-08 | 1.9e-15 | 2.1e-13 |
| 6 | TELPATHWAY | Telomerase is a ribonucleotide protein that adds telomeric repeats to the 3' ends of chromosomes. | AKT1, BCL2, EGFR, G22P1, HSPCA, IGF1R, KRAS2, MYC, POLR2A, PPP2CA, PRKCA, RB1, TEP1, TERF1, TERT, TNKS, TP53, XRCC5 | 15 | BCL2(1), EGFR(4), IGF1R(6), MYC(1), POLR2A(5), PPP2CA(1), PRKCA(1), RB1(17), TEP1(5), TERF1(4), TNKS(3), TP53(63), XRCC5(1) | 8095154 | 112 | 90 | 99 | 8 | 31 | 17 | 15 | 4 | 44 | 1 | 4.8e-06 | 2.4e-15 | 2.1e-13 |
| 7 | PLK3PATHWAY | Active Plk3 phosphorylates CDC25c, blocking the G2/M transition, and phosphorylates p53 to induce apoptosis. | ATM, ATR, CDC25C, CHEK1, CHEK2, CNK, TP53, YWHAH | 7 | ATM(7), ATR(6), CHEK1(1), CHEK2(2), TP53(63), YWHAH(1) | 4627970 | 80 | 72 | 67 | 3 | 21 | 18 | 9 | 5 | 27 | 0 | 0.000012 | 2.6e-15 | 2.1e-13 |
| 8 | PMLPATHWAY | Ring-shaped PML nuclear bodies regulate transcription and are required co-activators in p53- and DAXX-mediated apoptosis. | CREBBP, DAXX, HRAS, PAX3, PML, PRAM-1, RARA, RB1, SIRT1, SP100, TNF, TNFRSF1A, TNFRSF1B, TNFRSF6, TNFSF6, TP53, UBL1 | 13 | CREBBP(6), PAX3(2), PML(1), RARA(1), RB1(17), TNF(2), TNFRSF1A(1), TP53(63) | 5503411 | 93 | 81 | 80 | 2 | 26 | 17 | 7 | 4 | 38 | 1 | 2.6e-08 | 3.1e-15 | 2.1e-13 |
| 9 | WNTPATHWAY | The Wnt glycoprotein binds to membrane-bound receptors such as Frizzled to activate a number of signaling pathways, including that of beta-catenin. | APC, AXIN1, BTRC, CCND1, CREBBP, CSNK1A1, CSNK1D, CSNK2A1, CTBP1, CTNNB1, DVL1, FRAT1, FZD1, GSK3B, HDAC1, MADH4, MAP3K7, MAP3K7IP1, MYC, NLK, PPARD, PPP2CA, TCF1, TLE1, WIF1, WNT1 | 23 | APC(5), AXIN1(10), BTRC(1), CREBBP(6), CSNK2A1(1), CTNNB1(52), DVL1(1), FZD1(1), GSK3B(2), MAP3K7(1), MYC(1), NLK(1), PPARD(1), PPP2CA(1), TLE1(4), WIF1(1), WNT1(2) | 8805327 | 91 | 78 | 64 | 6 | 32 | 8 | 23 | 8 | 19 | 1 | 1e-05 | 3.1e-15 | 2.1e-13 |
| 10 | P53HYPOXIAPATHWAY | Hypoxia induces p53 accumulation and consequent apoptosis with p53-mediated cell cycle arrest, which is present under conditions of DNA damage. | ABCB1, AKT1, ATM, BAX, CDKN1A, CPB2, CSNK1A1, CSNK1D, FHL2, GADD45A, HIC1, HIF1A, HSPA1A, HSPCA, IGFBP3, MAPK8, MDM2, NFKBIB, NQO1, TP53 | 19 | ABCB1(5), ATM(7), CDKN1A(4), CPB2(2), HIF1A(1), IGFBP3(1), MDM2(1), NFKBIB(1), TP53(63) | 6314867 | 85 | 76 | 72 | 5 | 24 | 18 | 8 | 6 | 29 | 0 | 0.000032 | 5.1e-15 | 3.2e-13 |
Table 6. Get Full Table A Ranked List of Significantly Mutated Genesets (Excluding Significantly Mutated Genes). Number of significant genesets found: 0. Number of genesets displayed: 10
| rank | geneset | description | genes | N_genes | mut_tally | N | n | npat | nsite | nsil | n1 | n2 | n3 | n4 | n5 | n6 | p_ns_s | p | q |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | HSA00472_D_ARGININE_AND_D_ORNITHINE_METABOLISM | Genes involved in D-arginine and D-ornithine metabolism | DAO | 1 | DAO(5) | 206449 | 5 | 5 | 5 | 0 | 3 | 1 | 1 | 0 | 0 | 0 | 0.2 | 0.00078 | 0.48 |
| 2 | FOSBPATHWAY | FOSB gene expression and drug abuse | CDK5, FOSB, GRIA2, JUND, PPP1R1B | 5 | GRIA2(7), JUND(1), PPP1R1B(1) | 1097157 | 9 | 9 | 9 | 1 | 6 | 1 | 2 | 0 | 0 | 0 | 0.17 | 0.0044 | 0.78 |
| 3 | CYTOKINEPATHWAY | Intercellular signaling in the immune system occurs via secretion of cytokines, which promote antigen-dependent B and T cell response. | IFNA1, IFNB1, IFNG, IL10, IL12A, IL12B, IL13, IL14, IL15, IL16, IL17, IL18, IL1A, IL2, IL3, IL4, IL5, IL6, IL8, IL9, LTA, TNF | 20 | IFNB1(1), IFNG(2), IL12A(5), IL12B(1), IL16(2), IL18(2), IL4(1), IL6(2), LTA(1), TNF(2) | 2913639 | 19 | 19 | 19 | 2 | 7 | 1 | 7 | 2 | 2 | 0 | 0.079 | 0.0053 | 0.78 |
| 4 | ACHPATHWAY | Nicotinic acetylcholine receptors are ligand-gated ion channels that primarily mediate neuromuscular signaling and may inhibit neuronal apoptosis via the AKT pathway. | AKT1, BAD, CHRNB1, CHRNG, FOXO3A, MUSK, PIK3CA, PIK3R1, PTK2, PTK2B, RAPSN, SRC, TERT, TNFSF6, YWHAH | 13 | MUSK(4), PIK3CA(7), PIK3R1(2), PTK2(6), PTK2B(4), RAPSN(1), SRC(1), YWHAH(1) | 4891704 | 26 | 24 | 25 | 2 | 12 | 3 | 4 | 1 | 6 | 0 | 0.021 | 0.0059 | 0.78 |
| 5 | PTENPATHWAY | PTEN suppresses AKT-induced cell proliferation and antagonizes the action of PI3K. | AKT1, BCAR1, CDKN1B, FOXO3A, GRB2, ILK, ITGB1, MAPK1, MAPK3, PDK2, PDPK1, PIK3CA, PIK3R1, PTEN, PTK2, SHC1, SOS1, TNFSF6 | 15 | BCAR1(2), ITGB1(1), MAPK1(1), PDK2(1), PIK3CA(7), PIK3R1(2), PTK2(6), SHC1(2), SOS1(4) | 5556210 | 26 | 25 | 25 | 1 | 15 | 5 | 1 | 2 | 3 | 0 | 0.0028 | 0.0063 | 0.78 |
| 6 | TUBBYPATHWAY | Tubby is activated by phospholipase C activity and hydrolysis of PIP2, after which it enters the nucleus and regulates transcription. | CHRM1, GNAQ, GNB1, GNGT1, HTR2C, PLCB1, TUB | 7 | GNB1(1), HTR2C(4), PLCB1(7), TUB(1) | 2093699 | 13 | 13 | 13 | 1 | 3 | 3 | 3 | 0 | 4 | 0 | 0.15 | 0.0076 | 0.78 |
| 7 | VOBESITYPATHWAY | The adipose tissue of obese individuals overexpresses a key glucocorticoid-metabolizing enzyme, activating inactive circulating corticosteroids and inducing insulin resistance. | APM1, HSD11B1, LPL, NR3C1, PPARG, RETN, RXRA, TNF | 7 | LPL(1), NR3C1(2), RXRA(5), TNF(2) | 1684442 | 10 | 10 | 10 | 1 | 4 | 1 | 3 | 2 | 0 | 0 | 0.25 | 0.019 | 1 |
| 8 | FXRPATHWAY | The nuclear receptor transcription factors FXR and LXR are activated by cholesterol metabolites and regulate cholesterol homeostasis. | FABP6, LDLR, NR0B2, NR1H3, NR1H4, RXRA | 6 | LDLR(1), NR0B2(2), NR1H3(2), RXRA(5) | 1561481 | 10 | 10 | 10 | 0 | 5 | 3 | 0 | 2 | 0 | 0 | 0.05 | 0.022 | 1 |
| 9 | IFNGPATHWAY | IFN gamma signaling pathway | IFNG, IFNGR1, IFNGR2, JAK1, JAK2, STAT1 | 6 | IFNG(2), IFNGR1(2), JAK1(2), JAK2(4), STAT1(2) | 2415080 | 12 | 12 | 12 | 0 | 4 | 0 | 4 | 2 | 2 | 0 | 0.072 | 0.026 | 1 |
| 10 | IL18PATHWAY | Pro-inflammatory IL-18 is activated in macrophages by caspase-1 cleavage and, in conjunction with IL-12, stimulates Th1 cell differentiation. | CASP1, IFNG, IL12A, IL12B, IL18, IL2 | 6 | IFNG(2), IL12A(5), IL12B(1), IL18(2) | 906619 | 10 | 10 | 10 | 2 | 3 | 1 | 4 | 0 | 2 | 0 | 0.43 | 0.032 | 1 |
In brief, we tabulate the number of mutations and the number of covered bases for each gene. The counts are broken down by mutation context category: four context categories that are discovered by MutSig, and one for indel and 'null' mutations, which include indels, nonsense mutations, splice-site mutations, and non-stop (read-through) mutations. For each gene, we calculate the probability of seeing the observed constellation of mutations, i.e. the product P1 x P2 x ... x Pm, or a more extreme one, given the background mutation rates calculated across the dataset. [1]
In addition to the links below, the full results of the analysis summarized in this report can also be downloaded programmatically using firehose_get, or interactively from either the Broad GDAC website or TCGA Data Coordination Center Portal.